Active Drug Targeting by Receptor-Specific Ligands
Active targeting takes advantage of the over-expression of receptors on the tumor cell surface to achieve drug targeting by receptor-specific ligands. Specific interactions between the ligands on the surface of nanocarriers and receptors expressed on the tumor cells may facilitate nanoparticle internalization by triggering receptor-mediated endocytosis. CD Bioparticles have created a comprehensive platform to design these receptor-specific ligands based on our client need.
Introduction to Receptor-specific Ligands Used in Active Targeting
Recognizing that receptor-based active targeting of nanoparticles has the potential to be the optimal delivery strategy. Numerous targeting ligands have been employed to actively target nanoparticles including antibodies, folic acid, transferrin and so on.
Targeting of Antibodies Receptors
Antibodies were initially believed as “targeting missiles” which hit their specific biological targets. Not surprisingly, antibodies were among the first agents used for targeting nanovehicles to specific cell types based on surface antigens they presented. As targeting agents, antibodies have exceedingly high selectivity and binding affinity by virtue of the presence of two epitope binding sites in a single molecule. The specificity of antibodies lends particularly well to the active targeting of a variety of tumor types due to their ability to distinguish between healthy and cancerous cells and even amongst cancer cell types. Antibodies attached to the surfaces of nanoparticles target specific antigens present on the cell membrane. The use of antibodies as targeting moieties has been extensively investigated over the past decades and has resulted in numerous available treatments. There are various antibodies that have been found to be very effective and well-tolerable for the treatment of different malignancies. Monoclonal antibodies such as trastuzumab, gemtuzumab, and bevacizumab have been approved by FDA. The antibody-conjugated particles not only preferentially interacted with the cancerous cells but were also phagocytosed, which is ideal for the delivery of chemotherapeutic agents. Because antibodies have high specificity and affinity of antibody-antigen interaction, Ab mediated delivery is an attractive means for designing targeted nanocarriers. Besides whole Abs, antibody fragments such as Fab and scFv can be used for constructing targeted nanocarriers to overcome the size issue of antibody and achieve effective surface conjugation.
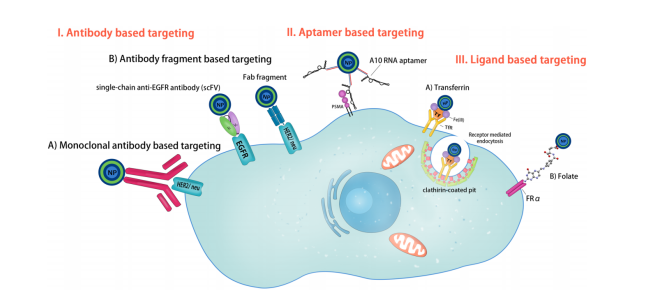
Figure 1. Schematic illustration of methods used for active targeting of nanoparticles. (Bazak, R., et al. Journal of cancer research and clinical oncology, 2015, 141(5), 769-784.)
Targeting of Folate Receptors (FRs)
Folate has been one of the most extensively utilized ligands for targeted drug delivery devices. The folate receptor, a 38 kDa glycosyl-phosphatidylinositol-anchored glycoprotein, is one of the most highly researched targets for cancer therapeutics. Folate Receptors are surface receptors, over expressed on various cancer cells such as lung, breast, brain, etc. Particles conjugated to folate receptor have greater chances of being internalized to a substantial extent, wherein the folate receptors are highly over-expressed. After internalized by the cell, these particles are introduced to the cytoplasm. The drug is then released by the nanoparticle in the cytoplasm of the tumor cell and proceed to interact with intracellular components. Folate modified particles display enhanced cellular uptake and circulation time as compared to unconjugated particles in folate-receptor positive cell lines. Folate ligands are widely used for targeting because they are inexpensive, nontoxic, nonimmunogenic, easy to conjugate to carriers, retain high binding affinity (KD~10-9), and are stable in storage and in circulation.
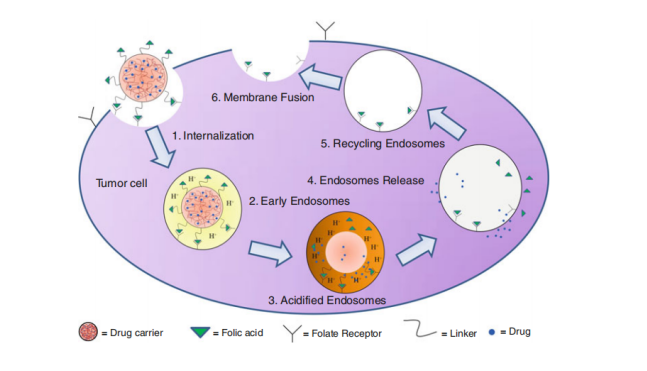
Figure 2. Mechanism of folate-mediated endocytosis for nanocarrier system. (Pawar, Prasad V., et al. Focal Controlled Drug Delivery. Springer, Boston, MA, 2014, 61-91.)
Targeting of Transferrin Receptors (TfRs)
Transferrin (Tf) is a serum iron Fe3+ carrier protein with mol wt about 80 kDa. TfRs (TfR1 and TfR2 or CD77) are receptors for transferrin (Tf) and are present on the cell surface. Due to the presence of an increased number of transferrin receptors on metastatic and drug resistant cells compared to normal healthy cells, transferrin is a very pertinent target for cancer therapeutics. Specifically, transferrin receptor (TfR) is highly upregulated on many tumor cells due to their higher demand for iron. TfR-mediated delivery can be obtained via the chemical conjugation of Tf or anti-TfR antibody or antibody fragments onto nanocarriers. Much like folate, when transferrin binds to TfRs. it initiates endocytosis and is internalized into the cellular cytoplasm, then iron dissociates. Transferrin-conjugated nanoparticles have been shown to inhibit cellular proliferation and tumor growth while participating in sustained release profiles and increased cellular uptake. And transferrin receptor targeting for cancer therapeutics has been used in human clinical trials with adriamycin, cisplatin, and diphtheria toxin.
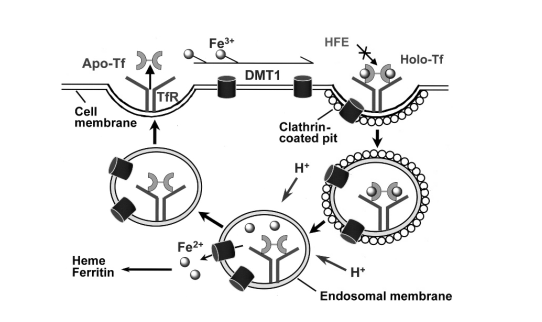
Figure 3. The transferrin cycle and the transferrin receptor-mediated cellular iron uptake. (Qian, Z. M., et al. Targeted drug delivery via the transferrin receptor-mediated endocytosis pathway. Pharmacological reviews, 2002, 54(4), 561-587.)
Our Receptor-Ligands Targeting Services
CD Bioparticles is specialized in the development of drug targeting strategy and customizing nanoparticles for drug delivery and targeting utilizing our core technologies. With our high-quality products and services, the efficacy of your drug delivery can be tremendously improved by receptor-specific ligands.
Our experienced scientists are proficient in providing products or synthesis services of targeting agents as antibodies (monoclonal antibodies and antibody fragments), folic acid and transferrin and their conjugation to nanoparticles according to your special needs. Types of nanoparticle and particle properties such as component molecular weight, surface charges and charge density, solubility, and hydrophobicity could be designed and engineered at your will.
Quotations and Ordering

References:
1. Vacchelli, E., Aranda, F., Eggermont, A., Galon, J., Sautes-Fridman, C., Zitvogel, L., Kroemer, G. and Galluzzi, L. Trial Watch: Tumor-targeting monoclonal antibodies in cancer therapy. Oncoimmunology, 2014, 3(1), e27048.
2. Zhao, X., Li, H. and Lee, R.J. Targeted drug delivery via folate receptors. Expert opinion on drug delivery, 2008, 5(3), 309-319.
3. Qian, Z.M., Li, H., Sun, H. and Ho, K. Targeted drug delivery via the transferrin receptor-mediated endocytosis pathway. Pharmacological reviews, 2002, 54(4), 561-587.





