Antimicrobial Lipid Nanoparticles Formulation, Development, and Function Evaluation
Introduction of Antimicrobial Lipid Nanoparticles
For years the rapid growth of antibiotic-resistant pathogens has severely jeopardized the health condition of human beings, yet the spread of such a threat continues to increase and has brought catastrophic consequences. Microbial pathogens have developed resistance mechanisms including: (i) decreased uptake and increased efflux of the drug through transmembrane efflux pumps; (ii) genetic alteration of the antibiotic targets with reduced affinity compared to the unaltered targets, (iii) production of enzymes which covalently modify or degrade the key structural motifs important for activity; and (iv) modification of membrane charge or sterol structure/composition.1
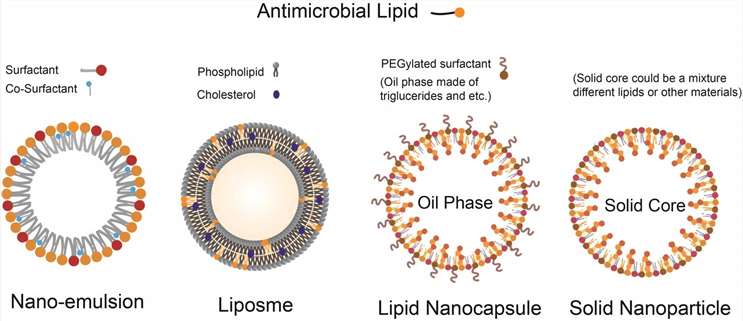 Figure 1. Illustration of the structure of nanoparticles composed of antimicrobial lipids.
Figure 1. Illustration of the structure of nanoparticles composed of antimicrobial lipids.
While the evolution of multidrug-resistance in harmful pathogens seems to be inevitable, modern science has pointed out appropriate answers, and microbial nanoparticles are currently one of the optimal solutions. They possess various traits such as drug protection from degradation, improved bioavailability, controlled release for a sustained concentration, specific targeting of diseased site, overcoming anatomical or cellular barriers, minimal toxicity, decreased emergence of drug-resistant, low cost, and scalable production.2 Lipid nanoparticles represents one of the best candidates.
Liposomes are composed of a bilayer membrane consisting of amphipathic phospholipids enclosing an interior space and have been researched intensively for antimicrobial drug delivery. Liposomes can exhibit antimicrobial properties, either alone or through encapsulation to enhance the antiviral efficacy of drugs. For example, Polyunsaturated ER liposomes (PERLs) significantly reduce viral secretion and infection for the treatment of HBV, HCV, and HIV infections. They act by the reduction of cholesterol which is essential for virus infectivity. Mareuil et al. 3 have compared the antiviral efficacy of SPC3 (a synthetic polymeric peptide) encapsulated in liposomes relative to free SPC3. Liposomal encapsulated SPC3 exhibited greater antiviral efficacy by more than ten and fivefold in HIV-infected C8166 T-cells and human peripheral blood lymphotcytes (PBLs), respectively.3 Besides, there are numerous papers reporting the successful application of lipid nanoparticles in delivering antimicrobial drugs. Table 1 is taken from reference 4 which lists out many of the well-studied antimicrobial lipid nanoparticles.4
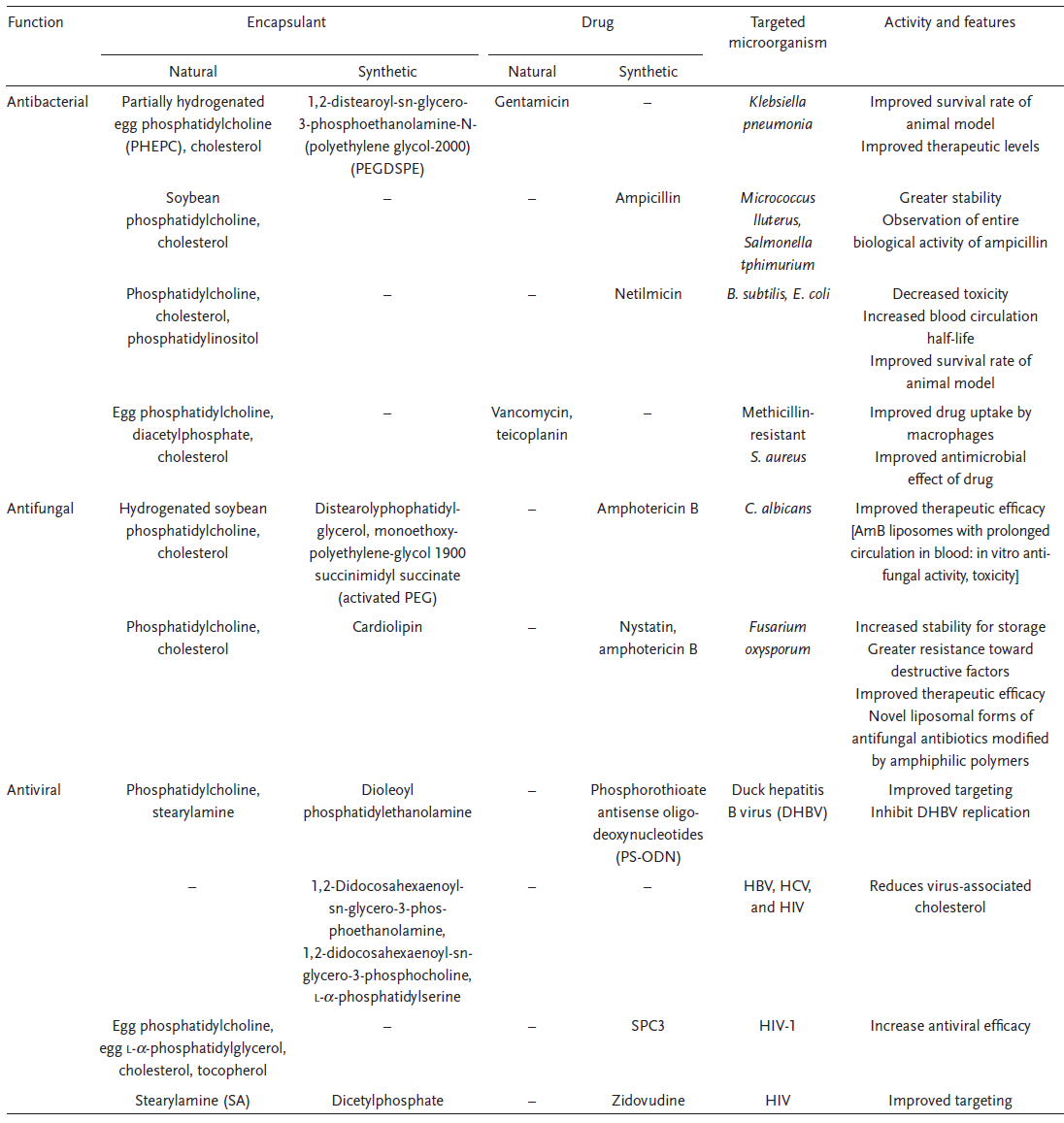
Table 1. Examples of antimicrobial liposome
Solid lipid nanoparticles (SLNs) composed of lipids that are solid at room temperature are great solutions as well. One of the advantages of SLNs is the comparably lower drug mobility in the solid lipid state compared to lipids in the oily phase, allowing better controlled release of drugs. Another advantage of SLNs is their stability in water and dermal creams. Table 2 is taken from reference 4 as well which lists out some major SLNs used in antimicrobial drug delivery.
In general, lipid nanoparticles are widely used in antimicrobial use, and have been proved to be effective.
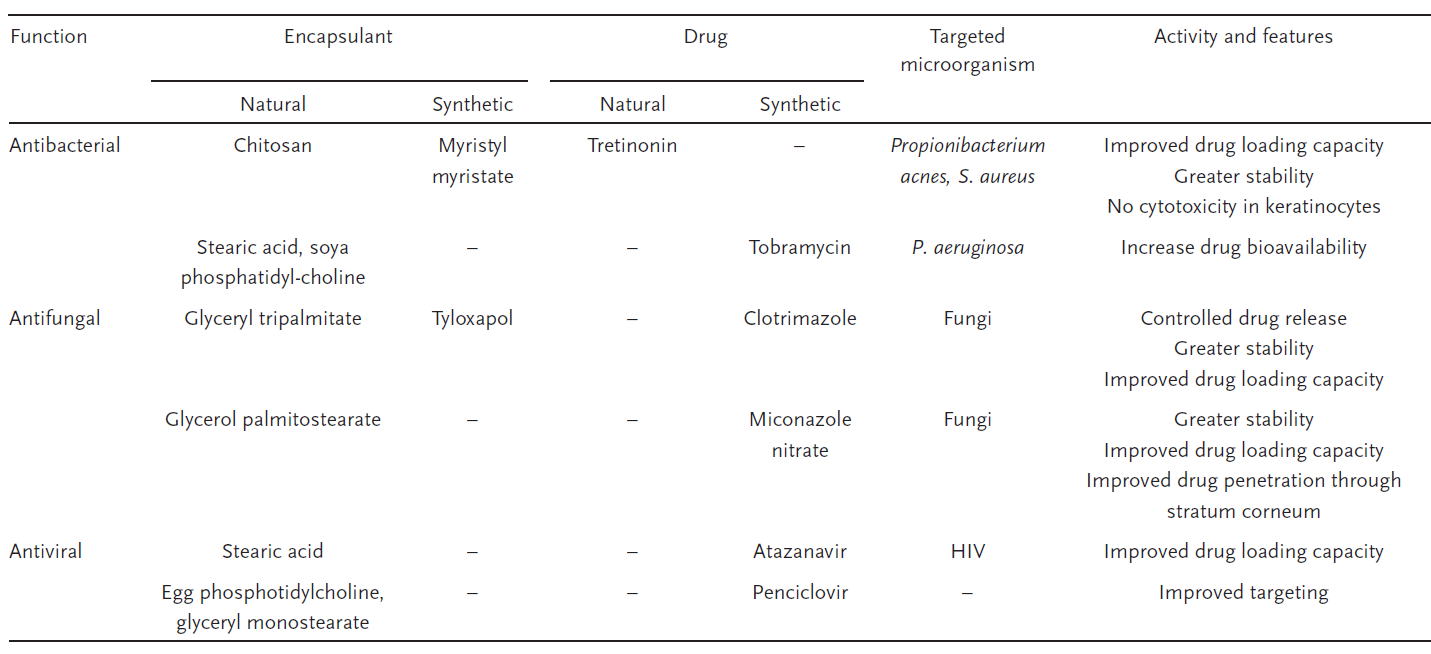
Table 2. Examples of antimicrobial SLNs
Challenges You Might Have:
-
The emergence of antibiotic resistance in bacteria results in the undruggable issues of recalcitrant infections
-
Prevalence of multidrug-resistance (MDR) in bacteria significantly increases morbidity and mortality of infected individuals
Key Antimicrobial Lipid Nanoparticles Services We Can Offer:
-
Design and screen the suitable liposomal cargos for antibiotics encapsulation and delivery
-
Optimize the liposomal cargos for your R&D purposes
-
Lipid library development for antimicrobial studies
-
Antimicrobial activity evaluation
Key Antimicrobial Lipid Nanoparticles Features:
-
Antibiotics delivery via lipid nanoparticles could result in a localized high concentration of the drug can be generated at the site of infection.
-
LNPs themselves, or in combination with a light source, can act as antimicrobial agents against resistant pathogens
-
Robust preparation methods (Table 3 is taken from reference 4)
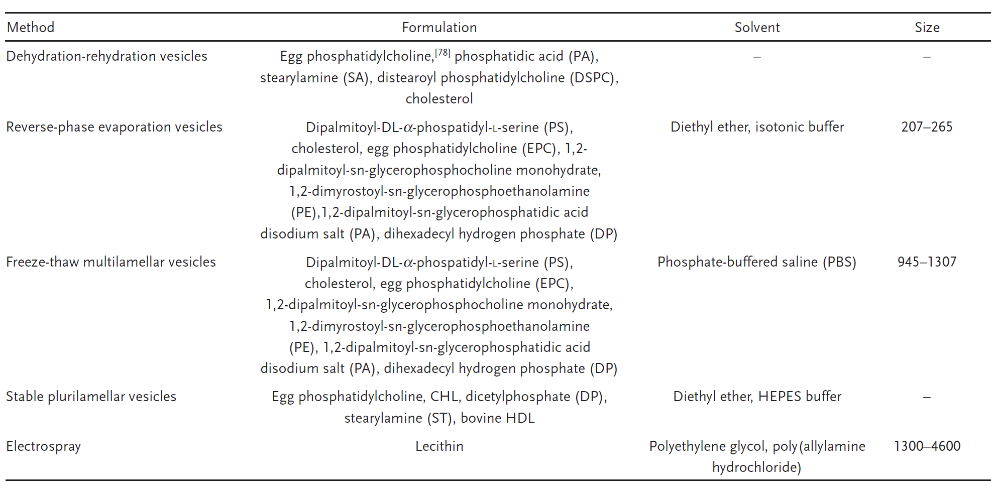
Table 3. Antimicrobial liposome preparation
Key Antimicrobial Lipid Nanoparticles Benefits:
-
The ability of LNP to penetrate eukaryotic cells
-
High drug loading capacity
-
The tunable properties of nanomaterials, particularly their surface functionalities, provide design spaces that can be fine- tuned to maximize the therapeutic effect while minimizing host toxicity.
References:
1. Matai I, Sachdev A, Dubey P, Kumar SU, Bhushan B, Gopinath P. Antibacterial activity and mechanism of Ag-ZnO nanocomposite on S. aureus and GFP-expressing antibiotic resistant E. coli. Colloids Surf B Biointerfaces. 2014 Mar 1;115:359-67.
2. Samiei M, Farjami A, Dizaj SM, Lotfipour F. Nanoparticles for antimicrobial purposes in Endodontics: A systematic review of in vitro studies. Mater Sci Eng C Mater Biol Appl. 2016 Jan 1;58:1269-78. doi: 10.1016/j.msec.2015.08.070. Epub 2015 Sep 4. PMID: 26478430.
3. de Mareuil J, Mabrouk K, Doria E, Moulard M, de Chasteigner S, Oughideni R, van Rietschoten J, Rochat H, De Waard M, Sabatier JM. Liposomal encapsulation enhances antiviral efficacy of SPC3 against human immunodeficiency virus type-1 infection in human lymphocytes. Antiviral Res. 2002 Jun;54(3):175-88. doi: 10.1016/s0166-3542(02)00002-5. PMID: 12062390.
4. Lakshminarayanan R, Ye E, Young DJ, Li Z, Loh XJ. Recent Advances in the Development of Antimicrobial Nanoparticles for Combating Resistant Pathogens. Adv Healthc Mater. 2018 Jul;7(13):e1701400. doi: 10.1002/adhm.201701400. Epub 2018 May 2. PMID: 29717819; PMCID: PMC7161883.


 Figure 1. Illustration of the structure of nanoparticles composed of antimicrobial lipids.
Figure 1. Illustration of the structure of nanoparticles composed of antimicrobial lipids.

