Buccal/ Sublingual Delivery
 CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy development. We are delicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial assay developers all around the world. One of our focus areas is buccal/ sublingual delivery which is defined as the administration of pharmaceutically active agents through the buccal/ sublingual mucosa to obtain local or systemic effects.
CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy development. We are delicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial assay developers all around the world. One of our focus areas is buccal/ sublingual delivery which is defined as the administration of pharmaceutically active agents through the buccal/ sublingual mucosa to obtain local or systemic effects.
Introduction to Buccal/Sublingual Delivery
Buccal medication administration refers to placing drugs between your gums and cheek and then drugs would dissolve and be absorbed into patient's blood, while sublingual administration refers to placing drugs under patient's tongue to dissolve and absorb into the blood. The dosage forms of them are usually liquid, semisolid, solid and sprays.
The oral mucosa is highly vascularized, so drugs can diffuse across the mucosa membranes, then enter the systemic circulation directly and will escape from hepatic metabolism. Besides, due to the higher overall permeability of the buccal and sublingual mucosa than other mucosa of the mouth, buccal/ sublingual delivery routes become the research focus for drug delivery. The continuous secretion of saliva also contributes to maintaining the relatively consistent and friendly physiological conditions for drug delivery in the oral cavity, which is better than the harsh environment of the GI tract. Differed from the secretions of the GI tract, saliva is a relatively mobile fluid and contains less mucin, limited enzymatic activity and barely no proteases. Moreover, there are fewer enzymes and lower enzyme activity in the buccal and sublingual regions, which favors the delivery of protein and peptide. Many studies have demonstrated that the predominant mechanism for buccal absorption is passive diffusion across lipid membranes via either the paracellular or transcellular pathways. To be specific, the hydrophilic nature of the paracellular spaces and cytoplasm is in favor of the delivery of hydrophilic drugs, while the lipophilic cell membrane favors delivery of lipophilic drugs.
Despite these advantages, buccal/ sublingual delivery has significant challenges for systemic drug delivery. Considering the release of drugs from the formulation to the buccal/ sublingual area and the pass through the mucosal to the systemic circulation, there are many physiological aspects of the oral cavity involving in this procedure such as pH, fluid volume, enzyme activity and the permeability of oral mucosa. In addition, the structure and turnover of the mucosal surface should be taken into consideration if you are designing an extended release drug delivery system in the oral cavity. Saliva flow would also lead to drug premature swallowing before effective absorption occurs through the oral mucosa. The saliva flow determines the time span of the release of drugs at the delivery site. Another major physiological barrier of buccal/ sublingual drug delivery is the drug permeability through the oral mucosa. The oral mucosa thickness and the composition of the epithelium are different between sites. Researchers have found that non-keratinized epithelia are more permeable to water than keratinized epithelia. At physiological pH, the mucus network would form a strongly cohesive structure to bind to the epithelial cell surface, which is vital in mucoadhesion for drug delivery systems. The turnover time for the buccal epithelium would affect the above systems too. The turnover time is about 3 to 8 days in the buccal epithelium while it's 30 days for the skin.
In recent years, researchers focus on studying particulates used for buccal mucosa delivery. Some of them have indicated that using nanoparticles as methods to formulate poorly water-soluble molecules would increase the dissolution of drugs and consequently obtain higher buccal bioavailability. ICNP-loaded ERL formulations were more effective in terms of film performance and insulin permeation through the human buccal mucosa, which indicates that it is a promising delivery system for buccal administration of a peptide such as insulin (Figure 1). Moreover, people also take buccal mucosa as a candidate site for vaccine administration. This area is easily reached and has numerous antigen presenting cells which can mediate immune responses to fight with local and systemic infection. To overcome the rapid clearance by saliva and tongue movement, researchers have applied some strategies to achieve buccal vaccination, namely supersaturation, nanoparticulates, nanofibers, iontophoresis, electroporation, and mucoadhesion.
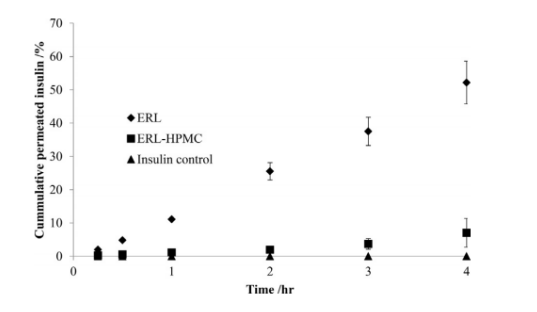
Figure 1. Cummulative insulin permeation of ICNP-loaded films and a control solution of insulin in PBS. (Morales, J. O., Huang, S., Williams, R. O., & McConville, J. T. (2014). Colloids and Surfaces B: Biointerfaces, 122, 38–45.)
Our Featured Services
CD Bioparticles is specialized in the development of drug delivery systems and customizing nanoparticles for drug delivery utilizing our core technologies. With our high-quality products and services, the efficacy of your drug delivery can be tremendously improved.
We offer custom synthesis of polymer microspheres and nanoparticles. Clients may select the material type, particle size, size distribution, color dye, fluorescent dye, and/or surface functional groups such as carboxyl or amine groups. We also encapsulate proteins, steroids, ligands, nucleic acids, and other drug molecules. Additionally, the surface coating of microspheres or nanoparticles with ligands, oligonucleotides, and other agents are available for clients to choose.
References:
1. Sattar, M., Sayed, O. M., & Lane, M. E. (2014). Oral transmucosal drug delivery – Current status and future prospects. International Journal of Pharmaceutics, 471(1-2), 498–506.
2. Patel, V. F., Liu, F., & Brown, M. B. (2011). Advances in oral transmucosal drug delivery. Journal of Controlled Release, 153(2), 106–116.
3. Paderni, C., Compilato, D., Giannola, L. I., & Campisi, G. (2012). Oral local drug delivery and new perspectives in oral drug formulation. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology, 114(3), e25–e34.


 CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy development. We are delicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial assay developers all around the world. One of our focus areas is buccal/ sublingual delivery which is defined as the administration of pharmaceutically active agents through the buccal/ sublingual mucosa to obtain local or systemic effects.
CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy development. We are delicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial assay developers all around the world. One of our focus areas is buccal/ sublingual delivery which is defined as the administration of pharmaceutically active agents through the buccal/ sublingual mucosa to obtain local or systemic effects.
