Cyclodextrin
Product Category
Cyclodextrin (CD) is a series of cyclic oligosaccharides produced from amylose under the action of cyclodextrin glycosyltransferase. It usually contains 6 to 12 D-glucopyranose units. Among them, molecules with 6, 7, and 8 glucose units are called α-, β-, and γ-cyclodextrins respectively. Currently, they have been widely used in the field of pharmaceutical preparations. Because the glycosidic bonds connecting the glucose units cannot rotate freely, cyclodextrins exhibit a slightly tapered ring structure rather than a cylindrical shape. The lower end of CD is composed of secondary hydroxyl groups at positions 2 and 3, while the upper end is composed of the hydrophilic primary hydroxyl group at position 6. The shielding effect of carbon-hydrogen bonds leads to the formation of a hydrophobic region in the cavity. Because cyclodextrin has a hydrophobic inner cavity and a hydrophilic outer structure, it can be co-assembled with various small molecules through van der Waals forces, hydrophobic interactions, and matching between host and guest molecules. At the same time, the multiple complex hydroxyl sites in CD also provide active sites for chemical modification of compounds. The primary hydroxyl group at position 6 has the lowest steric hindrance, the secondary hydroxyl group at position 2 is acidic, and the secondary hydroxyl group at position 3 is acidic. The hydroxyl group is inaccessible due to its active steric hindrance. Highly reactive reagents, such as trimethylsilyl chloride, react non-selectively with all hydroxyl groups, while bulky and poorly reactive reagents, such as tert-butyldimethylsilyl chloride, selectively bind to the 6-position hydroxyl group. The emergence of CD polymers has promoted the efficiency of drug delivery. CD polymers can be present as reactants in polymerization processes including reversible addition-fragmentation chain transfer, ring-opening polymerization, free radical polymerization, anionic polymerization, cationic polymerization, nitroxide-mediated free radical polymerization, and metathesis polymerization. These technologies can be applied to the synthesis of CD nanoparticles and enhance and expand the scope of drug applications.
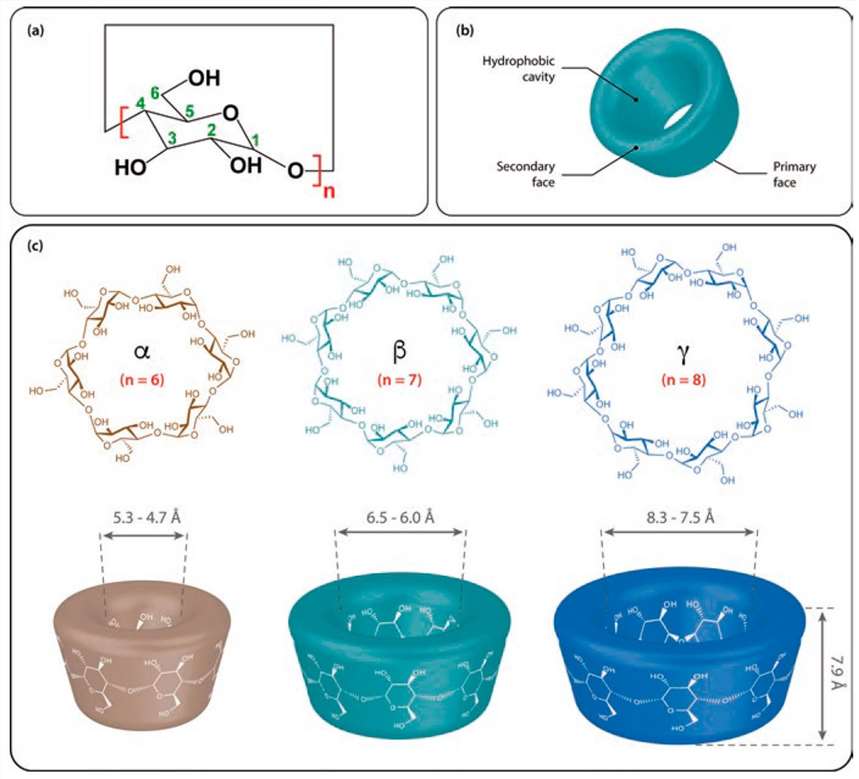 Figure 1. Cyclodextrin (CD) structures. (Jae-ung Lee, et al.; 2020)
Figure 1. Cyclodextrin (CD) structures. (Jae-ung Lee, et al.; 2020)
To this end, CD Bioparticles provides cyclodextrin drug delivery system to help solve the following challenges.
The challenges you might meet:
- Drugs lack targeting
- Drugs are toxic to non-target organs
- Drugs are easily degraded by gastric acid and other conditions after oral administration, making their bioavailability low.
- Drugs may be degraded by environmental influences in the body, thereby weakening their effectiveness.
- Drugs have low water solubility due to their specific chemical structure, making them difficult to absorb in the body
Cyclodextrin key features:
Cyclodextrin key benefits:
- By combining DNA or RNA with magnetic particles, directional guidance and transfection of cells can be achieved
- Cyclodextrin can encapsulate drugs and enhance their solubility in water. This is particularly important for drugs that have low water solubility due to their chemical structure.
- Cyclodextrin can improve the stability of drugs and protect them from the effects of light, heat, humidity and other factors in the environment.
- For oral administration, cyclodextrins can protect the drug from conditions such as gastric acid, preventing premature degradation in the stomach.
- Through appropriate encapsulation, cyclodextrin can reduce the toxicity of drugs to non-target organs, enable more targeted release of drugs into target tissues or cells, and reduce damage to normal tissues.
- By adjusting the structure of cyclodextrin, a specific drug delivery system can be designed to deliver the drug more effectively into the target tissue or cells and reduce the impact on other tissues.
- Cyclodextrin drug delivery systems can increase the local concentration of drugs, thereby increasing the drug's effect on specific targets and reducing the distribution range of the drug in the body.
Cyclodextrin application candidates:
- Cyclodextrins with hollow structures can form inclusion complexes with some hydrophobic or poorly soluble drugs, improving the water solubility and stability of these drugs
- It forms an inclusion complex with the drug to slowly release the drug in the body and prolong the action time of the drug
- For oral administration, it prevents drug degradation in gastric acid, thereby improving the bioavailability of oral administration
- It can slow down the absorption rate of drugs in the intestine and make the drugs absorbed more evenly
- Drug delivery to specific cells or tissues can be achieved
- Multiple drugs can be packaged at the same time to achieve multi-drug combination
- It can help drugs cross biofilms more easily and improve bio-accessibility


 Figure 1. Cyclodextrin (CD) structures. (Jae-ung Lee, et al.; 2020)
Figure 1. Cyclodextrin (CD) structures. (Jae-ung Lee, et al.; 2020)