Drug Localization Analysis
CD Bioparticles offers advanced drug localization study methods to help you analyze how drugs are distributed throughout the body and remain in the different amount of time especially when drug delivery systems are applied.
Drug Localization Analysis Introduction
Drug localization refers to the movement of a drug in the blood and various tissues of the body (for example, fat, muscle, and brain tissue) and the relative proportions of a drug in the tissues. The drug localization is usually varied and depends on several factors such as blood perfusion, tissue binding (since drug binding is linked to the lipid content), regional pH and cell membrane permeability. The localization of a drug in the body also depends on the extent to which the drug binds to proteins and tissues in the body. Only drugs that are unbound to proteins and other components in the blood are free to diffuse across the cell membranes into the tissues of the body. Drug carriers are designed to sustain drug circulation time and achieve drug targeting delivery, thus it becomes more important to investigate the drug distribution and localization with the effect of drug delivery bioparticles.
Drug Localization Analysis Methods
Plasma Protein Binding
Plasma protein binding refers to the degree to which medications attach to proteins within the blood. The extent of binding to plasma influences the way in which a drug distributes into tissues in the body. Extensive plasma protein binding also limits the amount of free compound available to access sites of action in the cell, and metabolism and elimination may be slower. Our team uses equilibrium dialysis to assess plasma protein binding. Compared with other methods such as ultrafiltration, it shows the lowest non-specific binding effects. We also deliver a value of the fraction of compound unbound to proteins as a reference.
Brain Tissue Binding
The main reason for different drug penetration characters is blood-brain barrier (BBB). BBB selectively transports of lipophilic compounds by active transport and slow down passive diffusion of water-soluble drugs. The drug penetration rate into CSF is determined mainly by the extent of protein binding, degree of ionization, and lipid-water partition coefficient of the drug. Our team can help you understand the extent of brain partitioning using our brain tissue binding assay.
Microsomal Binding
It has been recognized that drug sequestered in microsomes in vitro is presumed to be unavailable for direct interaction with metabolising enzymes, only the unbound substrate is free to interact with drug metabolizing enzymes in microsomes. As a result, illuminating the extent of microsomal binding can lead to a much better understanding of the relationship between in vitro drug metabolism data and in vivo pharmacokinetics, and can be useful in the investigation of hepatic clearance and drug-drug interactions. CD Bioparticles provides a high throughput microsomal binding assay to deliver a value of free fraction of compound in the microsomal. The data is collected by liquid chromatography-mass spectrometry(LC-MC/MC) after using a 96-well equilibrium dialysis unit to quickly and accurately determine the unbound fraction of drugs in the microsomal binding experiments.
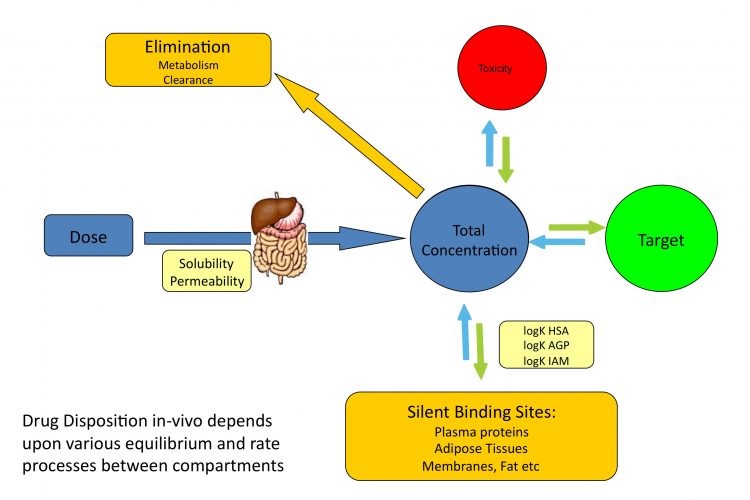
Figure.1 Drugs localization in various equilibrium between compartments. (Bio-mimetic chromatography to predict drug distribution in vivo - European Pharmaceutical Review)
CD Bioparticles offers a full set of service for drug localization analysis. For more detailed information, please feel free to contact us or directly send us an inquiry.
Quotations and Ordering

References:
1. Eddershaw, Peter J., Alan P. Beresford, and Martin K. Bayliss. ADME/PK as part of a rational approach to drug discovery. Drug Discovery Today. 2000, 5(9):409-414.
2. Dobrovolskaia, Marina A., et al. Interaction of colloidal gold nanoparticles with human blood: effects on particle size and analysis of plasma protein binding profiles. Nanomedicine: Nanotechnology, Biology and Medicine. 2009, 5(2): 106-117.
3. Wan, Hong, et al. High-throughput screening of drug− brain tissue binding and in silico prediction for assessment of central nervous system drug delivery. Journal of medicinal chemistry. 2007, 50(19): 4606-4615.
4. Austin, Rupert P., et al. The influence of nonspecific microsomal binding on apparent intrinsic clearance, and its prediction from physicochemical properties. Drug Metabolism and Disposition. 2002, 30(12): 1497-1503.



