Lamellarity Determination
In the development of liposome related drug carriers, the characterization of lamellarity is of the same essential importance as size analysis. CD Bioparticles offers custom services to measure the lamellarity of liposomes in different sizes using advanced microscopic techniques.
Lamellarity Determination Introduction
Lamellarity means the numbers of lipid bilayers in liposomes which influences the encapsulation efficiency and the drugs release kinetics. Furthermore, when liposomes are taken up or processed in the cell, the intracellular fate of liposome-drug complex is affected by the lamellarity. The liposome lamellarity has been affected largely by different lipid components or liposome preparation procedures. Therefore, liposomes lamellarity becomes an important parameter to characterize for quality control.
Lamellarity Analysis Methods
Liposome lamellarity is often accomplished by methods based on the visible or fluorescence signal change of lipid markers upon reagent addition. The results rely on the comparison of the total signal to the signal achieved from the reaction between the lipid markers and the specified reagents.
31P- nuclear Magnetic Resonance (NMR)
Magnetic resonance can detect the structure of liposomes. In 31P-NMR, the external shift reagents including Mn2+, Co2+, and Pr3+ are widely used. The Mn2+ ion is a paramagnetic ion that interacts with the phospholipids located in the outermost of the monolayer, leading to perturbations of the nuclear spin relaxation time. Broadened peaks and reduced signal strength in 31P-NMR spectrum can be quantified after data acquisition to calculate for liposome lamellarity. Under well-defined conditions, the analysis of liposomes by 31P-NMR is the presence of shift reagent in an elegant and accurate method gives useful information about the outer to inner phospholipids ratio. Our magnetic resonance platform could help our client to collect the information of lamellarity in unilamellar and multilamellar liposomes with the additional outside-inside distribution of phospholipids characterization.
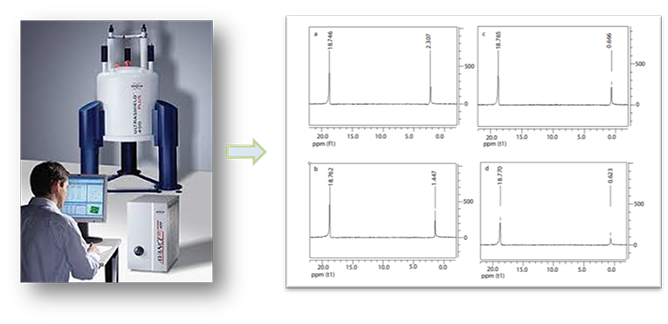
Figure 1. The data of 31P- nuclear magnetic resonance of liposomes. (Eyal Afergan, etc. J Bioanal Biomed (2010) 2: 125-131.)
Small Angle X-ray Scattering (SAXS)
Small angle X-ray scattering (SAXS) is considered as a good method evaluating vesicles lamellarity with high accuracy. Liposome dispersions in glass capillaries and curves are recorded with a camera equipped with a one-dimensional position sensitive detector. Blank scattering curves are obtained from the same capillaries filled with the liposome suspension solvent. Data is evaluated using the Indirect Fourier Transformation which provides the electron distance distribution p(r) calculated from the scattered intensity in the measured sample. p(r) gives the radial contrast profile of Δ p(r) in electron density relative to the mean value, which could be used to solve the internal structure of the scattering particles. This method provides accurate information on liposome lamellarity. Our collimation instruments platform could help our client to collect the liposome structural information of dimensions between 1 and 100 nm, and of repeat distances in partially ordered systems of up to 150 nm.
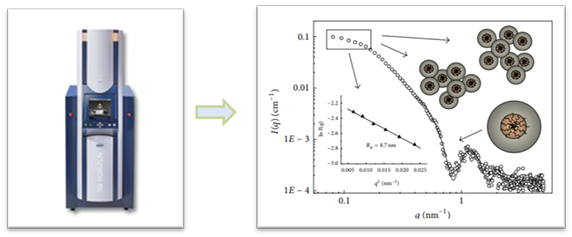
Figure 2. The data of small angle X-ray scattering of liposomes. (Domenico Lombardo, etc. Advances in Condensed Matter Physics (2015) DOI: 10.1155/2015/151683)
Cryo-electron Microscopy
To confirm the lamellarity results by an imaging method, the transmission electron microscopy (TEM) has been used. Cryo-EM specimens are prepared on carbon film coated copper grid. The data of lamellarity can be obtained by the image of Cryo-electron microscopy. Our high energy electrons (80keV-200keV) TEM platform could help our client to collect high resolutions images around 1-2Å, provide tailored testing service to characterize different types of liposomes in various sizes and numbers of the lipid bilayer.
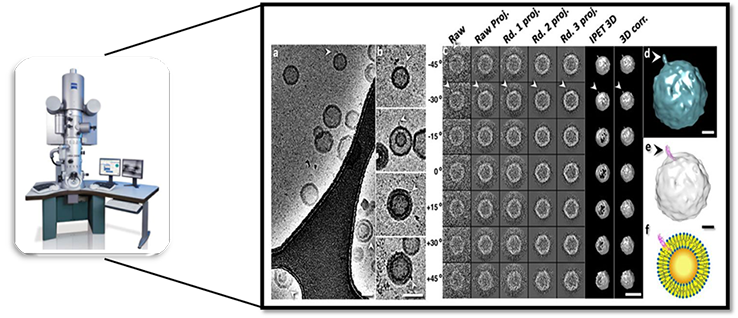
Figure 3. Transmission electron microscopy (TEM) image of liposomes (Masataka Chiba, etc. Biophysical Journal (2014) 107: 346–354).
Lamellarity determination featured services
In addition to the methods mentioned above, CD Bioparticles could also analyze liposome lamellarity using label-free differential interference contrast (DIC) microscopy, epifluorescence microscopy, and electron microscopy, etc. Our high cost-effective system provides rapid turnaround time for your samples, please contact us for a free consultation.
Quotations and Ordering

References:
1. M. Chiba, et al. Quantitative analysis of the lamellarity of giant liposomes prepared by the inverted emulsion method. Biophysical Journal. 2014, 107: 346-354.
2. C. Chen, et al. Analytical techniques for single-liposome characterization. Analytical Methods. 2013, 5: 2150–2157.
3. R. Suzuki, et al. Mono-cationic detergents play a critical role in the development of liposome-based gene vector via controlling its lamellarity. Nanoscale Research Letters. 2014, 16: 2227.
4. A. Laouini, et al. Preparation, Characterization and Applications of Liposomes: State of the Art. Colloid Sci. Biotechnol. 2012, 1( 2): 147–168.





