Lentiviral Vectors
CD Bioparticles uses 2nd or 3rd generation lentiviral vector packaging systems to provide customers with high-quality lentiviral vectors, ideal for gene therapy, construction of stable cell lines and scientific research applications. We have perfected a range of techniques and reagents that have significantly improved the titer, purity, and consistency of lentiviral vectors, ensuring reliable and consistent results. Moreover, we can provide you with various toxicological and biodistribution experimental research services to meet your specific scientific research requirements.
Introduction to Lentiviral Vectors[1]
Lentivirus belong to the retroviridae family. The gene integrating the lentiviral vectors can be expressed in long-term. The lentiviruses are able to express multiple genes on high-level in one location and have robust packaging capacity of tolerant up to 9 kb external gene. Lentivirus vector platform can achieve target tissue specifically by employing tissue-specific promoters to regulate transgene expression. In addition, applying endogenous miRNAs for post-transcriptionally regulation can reduce immune response against the foreign gene(s). Researcher and new drug developers combined CRISPR technique and next-generation vaccine design with lentivirus delivery system to large-scale screen the drugs and vaccine candidates.
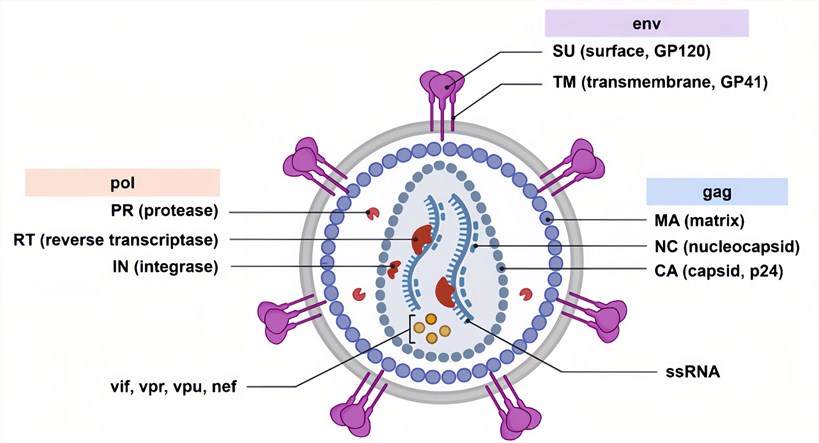 Figure 1. A schematic overview of the lentivirus HIV-1.
Figure 1. A schematic overview of the lentivirus HIV-1.
The Specific Features of Lentiviral Vectors Include:
-
Wide infection spectrum: Lentiviruses can efficiently infect both dividing and non-dividing cells, and are suitable for almost all cell lines, such as neuronal cells, hepatocytes, cardiomyocytes, tumor cells, endothelial cells, stem cells, etc.
-
Long-term stable expression: Lentiviruses can integrate exogenous genes into the host cell genome without losing them during cell division, and can achieve long-term stable expression of target genes.
-
Safe & Easy: The lentivirus uses a self-inactivating (SIN) replication-deficient virus strain to ensure safe operation.
The Applications of Lentiviral Vectors Include:
Gene therapy strategies based on lentivirus platform in vitro have had substantial advancements of treating genetic diseases, such as treatment of β-thalassemia, cerebral adrenoleukodystrophy, and treatment of inherited primary immune deficiency disorders.
-
Basic research: Now the retrovirus system has been widely used in gene overexpression in various cell lines, RNA interference, miRNA research and live animal experiments.
-
Construction of stable cell lines: Retroviruses can be used to create stable cell lines to express target proteins. Because they have the ability to integrate into the host genome in a stable fashion.
-
Gene therapy: Retroviruses have been used in a number of FDA-approved clinical trials. Retroviruses are suitable for dividing cells, such as stem cell-related research, neural stem cells and hematopoietic stem cells.
Our Lentiviral Vectors Featured Services:
CD Bioparticles has developed a series of proprietary technologies and reagents to improve the level of lentivirus packaging technology in terms of virus purity, titer, activity and consistency. Non-purified lentivirus can be used to transduce cells cultured in vitro (research-grade). The purified lentivirus can be used not only for cell transduction in vitro, but also for cell transduction in animals.
We Can Provide:
-
Lentiviral packaging service: Conventional gene overexpression / interference with lentiviral packaging
-
circRNA / LncRNA overexpression and interference
-
Customized CRISPR / Cas9 lentivirus
-
Lentiviral-based stable cell lines
CD Bioparticles pseudoviruses have the advantages of low cytotoxicity (ultracentrifugation purification process), no mycoplasma pollution, no endotoxin, no exogenous microorganisms, and no host nucleic acid residues.
-
in vitro and in vivo testing: localization studies, infection rate, gene expression assessment, immunogenicity assessment, in vivo distribution, in vivo metabolism, off-target evaluation, toxicological evaluation, etc.
We are committed to providing you with excellent services and products. Please contact us if you have questions.
Quotations and Ordering

Reference
-
Deng L, Liang P, Cui H.; Pseudotyped lentiviral vectors: Ready for translation into targeted cancer gene therapy? Genes & Diseases. 2022.


 Figure 1. A schematic overview of the lentivirus HIV-1.
Figure 1. A schematic overview of the lentivirus HIV-1.
