Nanoparticles for Cancer Treatment
CD Bioparticles is a leading manufacturer and supplier for various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers, and lipid system. All of them could be used in R&D and commercialization in a variety of application areas. We also have developed mature technology platforms for drug delivery, such as inorganic nanomaterials, biomacromolecules, polymeric and lipid system. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy customization. We are dedicated to providing the most comprehensive list of products and fit-for-purpose custom analysis services to academia as well as industrial researchers and assay developers all around the world.
For the last decade, researchers have paid significant interest to nanoparticles (NPs) due to their ability to overcome limitations in traditional chemotherapy of cancer treatment in drug delivery. Moreover, NPs can be custom synthesized to target specific cancer tissues and carry both drugs and imaging probes at the same time. The first clinical trial of NPs for cancer treatment drug delivery can be dated back to the middle 1980s while the first therapeutic NPs entered the pharmaceutical market were in 1995. From then on, numerous new NPs for cancer drug delivery have been approved or developed because of their obvious advantages including enhancing the solubility of hydrophobic drugs, prolonging circulation time, minimizing nonspecific uptake, reducing off-target and side effects, improving intracellular penetration, and ability of specific cancer targeting.
Nanoparticles in Cancer Treatment
As the third leading cause of mortality all over the world, cancer is a disease due to abnormal cell growth. In 2015, it caused 8,800,000 deaths. To kill the cancer cells, we currently use chemotherapeutic drugs in chemo or radiation therapy. However, these treatments usually bring several side effects (e.g. damaging the surrounding healthy tissues), and may delay in diagnosis or have a high incidence of relapse, which lowers the survival rates. Using a nanoparticle-based drug delivery approach to treat cancer cells can overcome the limitations of conventional treatment methodologies by providing simultaneous diagnosis and treatment. Although gold, silver, iron, copper, cobalt, platinum, etc. have been applied in NPs and nanomaterials, gold NPs have been the No. 1 choice for researchers in the biological and pharmaceutical field because of their unique optical and Surface Plasmon Resonance (SPR) properties. Moreover, the ability to utilize NP features shows many possibilities to apply these NPs in drug delivery of cancer treatment as image contrast agents and for diagnostic purposes.
Polymers in Cancer Treatment
Polymer materials are known for their versatile and customized properties and play a crucial role in the field of cancer treatment. These materials can be designed as drug carriers to protect them when the drug passes through the complex systems of the human body and delivers it directly to the tumor site. By acting as a carrier, high-molecular materials enhance drug stability, control release rates and target malicious cells more precisely. This has led to their clinical application in a variety of cancer treatments, offering the potential to improve therapeutic effects and reduce adverse reactions.
Liposomes in Cancer Treatment
Liposomes, a tiny spherical structure made up of lipids similar to cell membranes, have shown tremendous potential as carriers for cancer treatment. Their unique structure enables them to pack a wide range of drugs, from traditional chemotherapy drugs to updated targeted therapeutic drugs. By capturing these drugs within their double layer of lipid, liposomes can protect them from degradation and ensure controlled release. This controlled delivery not only enhances the effectiveness of the drug against the tumor, but also minimizes damage to healthy tissues. The FDA has recognized these benefits and approved certain liposome-based treatments for specific types of cancer, marking an important step forward in personalized and less toxic cancer treatments.
Other Carriers in Cancer Treatment
Beyond polymers and liposomes, a spectrum of other carriers has also emerged as essential tools in the fight against cancer. For example, micelles, metallic nanoparticles, dendrimers, carbon nanotubes, silica nanoparticles, lipid-based nanocarriers, polymeric micelles, hybrid nanocarriers, quantum dots, and magnetic nanoparticles. Additionally, protein-based carriers, such as antibodies, offer a targeted approach by homing in on specific proteins present on cancer cell surfaces. This level of specificity reduces collateral damage to healthy cells and is paving the way for more tailored and effective treatments. As these carriers navigate the path from research to clinical trials and FDA approvals, they hold promise for shaping the future landscape of cancer therapy.
Successful cases of nanoparticle-based delivery of cancer therapeutics:
-
Abruafinib (Tafinlar®) and Dabrafenib (Tafinlar®):
These are kinase inhibitors used to treat specific types of melanoma. They have been formulated into nanoparticles for improved drug delivery and efficacy.
-
Paclitaxel (Abraxane®):
Paclitaxel, a chemotherapy drug, has been formulated using albumin-based nanoparticles in the drug Abraxane®. It's used for the treatment of breast cancer, pancreatic cancer, and non-small cell lung cancer.
-
Irinotecan Liposome (Onivyde®):
Irinotecan, a chemotherapy drug, has been encapsulated in liposomes to create Onivyde®. It's approved for the treatment of metastatic pancreatic cancer in combination with other drugs.
-
Cerliponase Alfa (Brineura®):
Although not for traditional cancer treatment, this enzyme replacement therapy uses nanoparticles to treat a specific form of pediatric brain cancer known as neuronal ceroid lipofuscinosis type 2 (CLN2) disease.
-
Daunorubicin-Cytarabine Liposome (Vyxeos®):
Vyxeos® is a combination chemotherapy encapsulated in liposomes for the treatment of specific types of acute myeloid leukemia (AML).
-
Calaspargase Pegol-mknl (Asparlas®):
This is a nanoparticle-encapsulated version of the enzyme L-asparaginase, used as a component of multi-agent chemotherapy for the treatment of acute lymphoblastic leukemia (ALL) in pediatric and young adult patients.
The application of targeted NPs to deliver chemotherapeutic agents in cancer treatment possesses many advantages including improving drug/gene delivery and overcoming many disadvantages associated with traditional chemotherapy:
-
Through either passive targeting or active targeting, NPs can enhance the intracellular concentration of drugs/genes in cancer cells (Figure 1) and avoid toxicity in normal cells at the same time.
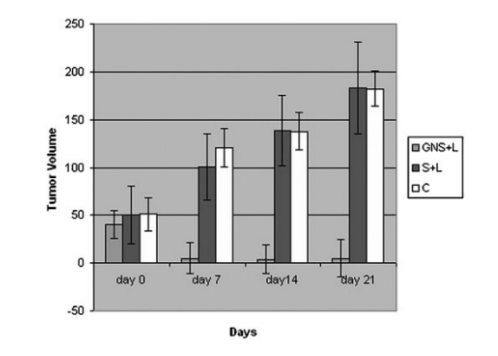
Figure 1. Tumor growth of human prostate cancer tumors in mice. C, control; GNS, gold nanoshells; L, laser; S, sham treated. (Jain, S., Hirst, D. G., & O’Sullivan, J. M. (2012). Gold nanoparticles as novel agents for cancer therapy. The British Journal of Radiology, 85(1010), 101–113)
-
The targeted NPs can be custom synthesized as a pH-sensitive or temperature-sensitive carrier. The pH-sensitive drug delivery system is ideal for delivering and releasing drugs within the more acidic microenvironment of the cancer cells (Figure 2), while the temperature-sensitive system combined with chemotherapy and hyperthermia can carry and release drugs to tumor region having temperature changes.
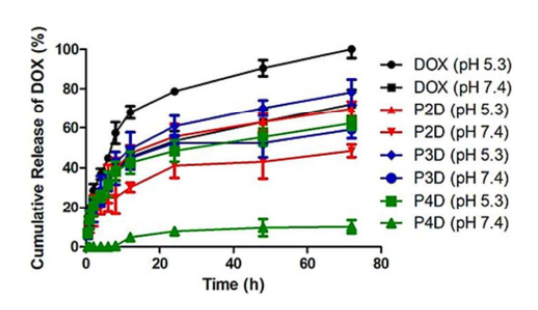
Figure 2. Cumulative drug release graph of free DOX at pH 5.3 and pH 7.4 and AuNP/Polymer-DOX bioconjugates at pH 5.3 and pH 7.4 for 72 h at 37 °C. DOX: doxorubicin. (Yilmaz, G., et al. (2018). pH responsive glycopolymer nanoparticles for targeted delivery of anti-cancer drugs. Molecular Systems Design & Engineering, 3(1), 150–158)
-
The targeted NPs can be modified or functionalized so that their toxicity is reduced. For example, modifying NPs surface chemistry could result in lower toxicity and immunotoxicity (Figure 3).
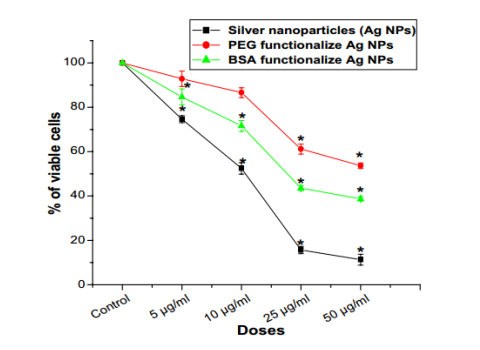
Figure 3. In vitro cytotoxicity of bare silver nanoparticles and surface modified silver nanoparticles against PBMCs. n = 6; values are expressed as mean ± SEM. Asterisk Indicates the significant difference as compared to control group. (Das, B., et al. (2017). Surface modification minimizes the toxicity of silver nanoparticles: an in vitro and in vivo study. JBIC Journal of Biological Inorganic Chemistry, 22(6), 893–918)
Like other new technologies, targeted NPs for cancer treatment face many challenges too. NPs may change the stability, solubility, and pharmacokinetic properties of the carried drugs and other aspects such as the shelf life, aggregation, leakage, and toxicity of materials used to make nanoparticles limit their use too. For example, poly (lactic-co-glycolic acid) (PLGA) is with low toxicity and will degrade quickly which means there is not enough time to circulate in tissues for sustained drug delivery. On the other hand, although carbon nanotubes and quantum dots are durable and can exist in the patient's body for weeks, months or even longer, their potentially toxic limits their use for repeated treatments. As for silica NPs, researchers developed them for drug delivery to treat cancer but also worry about the potential risks for introducing new materials in the human body. In addition to making efforts to develop new material and select appropriate materials for each specific treatment, there are other factors that should be considered to design better targeted NPs. These factors are the particle size, shape, sedimentation, drug encapsulation efficacy, drug release profiles, distribution in the body, circulation, and cost. For example, the clearance rate of very small NPs may be high and most of these NPs may end up in patient's liver and spleen, leading to the impractical and ineffective use of the targeted NPs. However, larger NPs are too big to go through small capillaries for drug delivery. As a result, it is vital to select the right materials and particle size of targeted NPs for cancer treatment.
Our Featured Services
CD Bioparticles is specialized in the development of drug delivery systems and customizing nanocarriers for drug delivery utilizing our core technologies. With our high-quality products and services, the efficacy of your drug delivery can be tremendously improved.
We offer custom synthesis of polymer microspheres and nanoparticles for cancer treatment. Clients may select the material type, particle size, size distribution, color dye, fluorescent dye, and/or surface functional groups such as carboxyl or amine groups. We enable you to make wise decisions on all components of the antibody-drug conjugate (ADC) structure so that the optimal candidate is identified for your target of interest in terms of stability, off-target toxicity, and efficacy. Besides, methodologies developed to produce the ADC are simple, scalable and reproducible and ultimately suitable for large-scale manufacture.
References
-
Singh, P., Pandit, S., Mokkapati, V. R. S. S., Garg, A., Ravikumar, V., & Mijakovic, I. (2018). Gold Nanoparticles in Diagnostics and Therapeutics for Human Cancer. International Journal of Molecular Sciences, 19(7), 1979.
-
Nguyen KT (2011) Targeted Nanoparticles for Cancer Therapy: Promises and Challenges. J Nanomedic Nanotechnol, 2, 103e.
-
Tian H, et al.; Enhancing the therapeutic efficacy of nanoparticles for cancer treatment using versatile targeted strategies. J Hematol Oncol. 2022, 15(1):132.
-
Mundekkad D, Cho WC. Nanoparticles in Clinical Translation for Cancer Therapy. Int J Mol Sci. 2022, 23(3):1685.
-
Szewczyk OK, et al.; An Overview of the Importance of Transition-Metal Nanoparticles in Cancer Research. Int J Mol Sci. 2022, 23(12):6688.




