Nanoparticles for Cardiovascular Disease Treatment
CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. We also have developed mature technology platforms for drug delivery, such as inorganic nanomaterials, biomacromolecules, polymeric and lipid system. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy. We are dedicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial researchers and assay developers all around the world.
Vasodilators, statins, and thrombolytic drugs are often directly delivered into the bloodstream to treat cardiovascular disease (CVD). However, these drugs may cause systemic toxicity problems. So, people have paid more attention to nanotechnology-based approaches which could offer a route to deliver formulations (e.g. therapeutic agents) stably and bypass their detrimental systemic side effects. The main goals of nanoparticle drug delivery system are to improve the safety and efficacy by passive or active targeted delivery and to improve the solubility of drug candidates.
Nanoparticles in Cardiovascular Disease Treatment
According to the WHO data, cardiovascular diseases are the main cause of death all over the world. 17.7 million people died of CVDs in 2015 and this figure may increase to 23.6 million in 2030. CVDs consist of coronary heart disease, deep vein thrombosis, pulmonary embolism, myocardial infarction (MI), etc. Although all these diseases could result in ischemia and tissue death, MI and heart failure are still the No.1 cause of mortality. For the past two decades, a number of nanotechnologies have been developed for biomedicine to treat CVDs, including liposomes, nanoparticles, micelles, and dendrimers. In addition to cardiovascular medicine, the therapeutic applications of nanotechnology within medicine are also focused on cancer treatment and diagnosis and antimicrobial resistance.
-
Liposomes
Liposomes are 50-200 nm in size and form phospholipid bilayer structures within which encapsulate aqueous cores. Because they simultaneously have characteristics of hydrophilicity and hydrophobicity, liposomes are ideal for effective drug delivery systems (Figure 1). According to the size and number of bilayers, liposomes can be classified into three types: small unilamellar vesicles (SUV) with a single lipid bilayer; large unilamellar vesicles (LUV); multilamellar vesicles (MLV) with numerous bilayers. Generally, variable size, preparation method, surface charge, and lipid composition are all factors influencing the performance of liposomes. As observed in other liposomal drug delivery applications, the use of liposomes has some disadvantages. There is limited long-term stability of liposome drug delivery system, thus leading to premature drug release. Moreover, this system is usually more expensive and less effective than polymer-based systems due to the increased excipient-to-drug ratios.
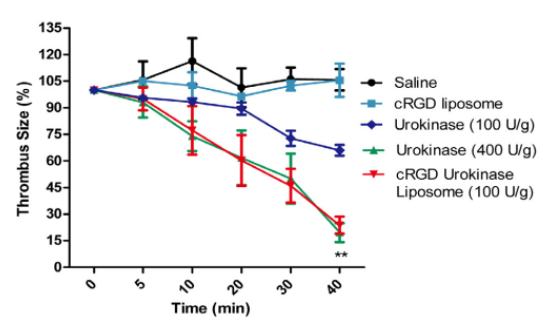
Figure 1. The real-time thrombolysis results of the cyclic RGD (cRGD) liposomes encapsulating urokinase in mouse mesenteric vessels. The images were taken at time 0 min, 5 min, 10 min, 20 min, 30 min and 40 min post the formation of thrombus. The reduction of thrombus size for saline, free urokinase and the cRGD urokinase liposomes respectively. The thrombus size was defined as or equaled to the area of TRITC fluorescent signals analyzed by a digital image analyzing software. The thrombus size at time 0 min was set as 100%. The experiments were repeated 5 times and the results were represented as mean ± standard deviation (SD). Student’s T test was used to compare the difference between group free urokinase (100 U/g) and cRGD urokinase liposomes (100 U/g). *P < 0.05, **P < 0.01. (Zhang, N., et al. (2018). Cyclic RGD functionalized liposomes encapsulating urokinase for thrombolysis. Acta Biomaterialia, 70, 227–236)
-
Polymeric Nanoparticles
Polymeric nanoparticles are manipulable materials and have the potential of reabsorption in the body, which makes them attract more researchers' attention in the nanomedicine field. The site of action and the choice of minimally invasive method to get them into systemic circulation determine the delivery route of polymeric nanoparticles. Although intravenous injection is most commonly used, oral, mucosal, dermal, or transdermal route are available to deliver polymeric nanoparticles as well. In addition, polymer-based systems are more cost-effective and easier to produce and scale up than liposomes with long stability profiles. However, researchers have found chronic toxicity in many non-biodegradable polymeric materials including poly(methyl methacrylate) (PMMA), poly(acrylamide), poly(styrene), and poly(acrylates). So, they have changed their focus of drug delivery system to studying biodegradable polymers such as poly(lactide) (PLA), poly(lactide-co-glycolide) copolymers (PLGA), and poly(ε-caprolactone) (PCL).
Gelated nanoparticles are the least common polymer-based particles. With increased polymer concentration, these nanoparticles incorporated with drugs could form into hydrogel matrices. These systems are with better long-term stability and available for longer retardation of drug release. Oh, et al. found that the MI group and the NP group showed decreased systolic and diastolic functions. Moreover, the VEGF+NP group and the VEGF+NP/ GEL group showed a significantly increased cardiac function. A more significant improvement was observed in the VEGF+NP/GEL group. However, the VEGF+NP/GEL group didn’t show the improvement in the arterial elastance (Figure 2).
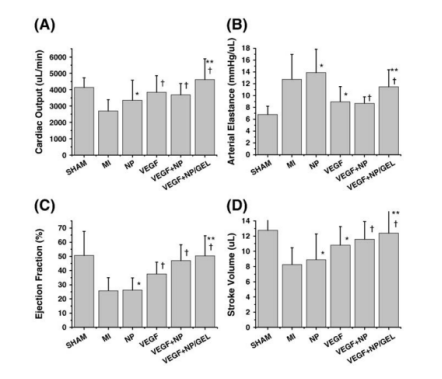
Figure 2. Functional evaluation of an ischemic heart. (A) Cardiac output, (B) arterial elastance, (C) ejection fraction, and (D) stroke volume. SHAM: a sham operation group; MI: a myocardial infarction group; NP: a group injected with core/shell nanoparticles without any VEGF (vascular endothelial growth factor); VEGF: a group injected with 5 μg of soluble VEGF; VEGF+NP: a group injected with core/shell nanoparticles loaded with the VEGF (5 μg); VEGF+NP/GEL: a group injected with gel composed of the VEGF-loaded core/shell nanoparticles. (Oh, K. S., et al. (2010). Temperature-induced gel formation of core/shell nanoparticles for the regeneration of ischemic heart. Journal of Controlled Release, 146(2), 207–211)
-
Micelles
Micelles are composed of lipids and other amphiphilic artificial molecules (e.g. polymers). In aqueous solution, micelles are self-assembling to form a monolayer with a hydrophobic phase inside, which makes them appropriate to incorporate hydrophobic therapeutic agents. Compare to liposomes, micelles are usually 10-100 nm in diameter and more controllable to add targeting ligands or tracking molecules. Moreover, micelles may be with lower systemic toxicity and are more cost-effective.
-
Dendrimers
The dendrimers are multibranched, three-dimensional architecture, and are coupled with low polydispersity and great functionality. Compared to other nanotechnologies, dendrimers are appropriate for delivery of non-viral vectors for therapeutic indications due to their high solubility, greater stability, minimal immunogenicity, and ability to facilitate effective delivery of drug molecules, DNA, and RNAs. One drawback to dendrimers is that they usually have either highly cationic or highly anionic, which may lead to toxicity problems.
Nanoparticles for Cardiovascular Disease Diagnostic Imaging
Nanoparticles have high potential in medical imaging because of their good bioavailability profiles, the versatility of use, and ease of manipulation. Meanwhile, nanoparticles are with adaptable qualities which allow for generating multimodal and multifunctional imaging vehicles with modifiable features. Researchers can integrate contrast materials, ligands, and functional groups to nanoparticles. For example, gold nanoparticles show great promise in both cardiovascular medicine and oncology imaging. They can be excreted by the kidney. When attaching PEG chains to the nanoparticle surface, the elimination rate of gold nanoparticles through glomerular filtration can be modified and the circulation time and hydrophilicity of these particles will be increased too.
Nanoparticles for Cardiovascular Disease Treatment
The potential use of nanoparticles in treating cardiovascular disorders, which include conditions affecting the heart and blood arteries, has drawn attention in the realm of medical research. These minuscule particles, which are frequently smaller than viruses, are designed to deliver drugs or other therapeutic agents to certain organ targets, opening up a new avenue for the treatment of many ailments, including cardiovascular ones.
The direct delivery of medications to atherosclerotic plaques, which are fatty deposits that accumulate in arteries and constrict blood vessels, increasing the risk of heart attacks and strokes, is one significant application of nanoparticle technology. Targeting these plaques with nanoparticles that deliver drugs that lessen inflammation, support plaque stability, or even dissolve the plaque itself may help to avoid or minimize the risk of cardiovascular events.
In the area of regenerative medicine, nanoparticles have another interesting application. Researchers are looking into the idea of directly delivering stem cells or growth hormones to injured heart tissue using nanoparticles. With the potential to undo the harm caused by heart attacks and enhance overall heart function, this strategy tries to promote the regeneration of healthy heart cells.
Additionally, the effectiveness of conventional cardiovascular therapy can be increased by using nanoparticles. For instance, they can improve the delivery of conventional medications, which may be less effective due to poor solubility or quick bodily clearance. These medications' stability and bioavailability can be improved by encasing them inside nanoparticles, enabling more efficient therapy of disorders like hypertension or arrhythmias.
Cases Approved by FDA or in Clinical Research
There are currently no FDA-approved medications that are intended to be used as nanoparticles to treat cardiovascular disorders. Nevertheless, various nanoparticle-based medication delivery methods were being researched, and some had even reached the stage of clinical testing. Here are a few instances:
Statins made using nanoparticles: Statins are frequently used to decrease cholesterol and lower the risk of cardiovascular events. To increase the delivery of statins to atherosclerotic plaques, scientists were investigating the use of nanoparticles. The targeting of statins to the plaque site may be improved by these nanoparticles, which would increase their efficiency in lowering plaque inflammation and stabilizing the plaques.
Stents with nanoparticle coatings: Stents are frequently used to treat blocked or constricted arteries by increasing blood flow. To avoid restenosis (re-narrowing of the artery) following stent implantation, researchers were aiming to create stents coated with nanoparticles that deliver medications. Anti-proliferative medications might be gently released by these nanoparticles, aiding in the reduction of uncontrollable tissue growth at the stent site.
Toxicity of Nanoparticles
Although the utilization of nanoparticles has many advantages in increasing the efficacy of CVD treatment, it is important to fully study cytotoxic effects when using these nanoparticles. Because nanoparticles are mobile in circulation throughout the patient's body and thus the toxicity cannot be ignored. Recent researches have indicated that when applying different varieties of nanoparticles to the cardiovascular system, specific cytotoxic effects will occur. For instance, Nemmar, et al. found that ultra-small superparamagnetic iron oxide nanoparticles (USPIO) (composed of magnetite Fe3O4 and <1.0% stabilizing ligands of poly ethylene glycol) have adverse effects on myocardium including thrombotic response in vivo, platelet aggregation in vitro with whole blood, DNA damage, and increase of cardiac oxidative stress. So, before the wide-spread use of Fe-based nanoparticles, it's vital to carefully study their impact on patient's health and the environment through the lifetime.
Our Featured Services
CD Bioparticles is specialized in the development of drug delivery systems and customizing nanocarriers for drug delivery utilizing our core technologies. With our high-quality products and services, the efficacy of your drug delivery can be tremendously improved.
We offer custom synthesis of polymer microspheres and nanoparticles for cardiovascular disease diagnostics and treatment. Clients may select the material type, particle size, size distribution, color dye, fluorescent dye, and/or surface functional groups such as carboxyl or amine groups. We enable you to make wise decisions on all components of the antibody-drug conjugate (ADC) structure so that the optimal candidate is identified for your target of interest in terms of stability, off-target toxicity, and efficacy. Besides, methodologies developed to produce the ADC are simple, scalable and reproducible and ultimately suitable for large-scale manufacture.
References
-
Suarez, S., Almutairi, A., & Christman, K. L. (2015). Micro- and nanoparticles for treating cardiovascular disease. Biomaterials Science, 3(4), 564–580.
-
Chandarana, M., Curtis, A., & Hoskins, C. (2018). The use of nanotechnology in cardiovascular disease. Applied Nanoscience, 8, 1607.
-
Jiang, W., Rutherford, D., Vuong, T., & Liu, H. (2017). Nanomaterials for treating cardiovascular diseases: A review. Bioactive Materials, 2(4), 185–198.
-
Nemmar, A., Beegam, S., Yuvaraju, P., Yasin, J., Tariq, S., Attoub, S., & Ali, B. H. (2015). Ultrasmall superparamagnetic iron oxide nanoparticles acutely promote thrombosis and cardiac oxidative stress and DNA damage in mice. Particle and Fibre Toxicology, 13(1).
-
Duivenvoorden R, et al.; A statin-loaded reconstituted high-density lipoprotein nanoparticle inhibits atherosclerotic plaque inflammation. Nat Commun. 2014, 5:3065.
-
Zhang ZQ, et al.; Advances in coatings on magnesium alloys for cardiovascular stents - A review. Bioact Mater. 2021, 6(12):4729-4757.



