Nanoparticles for Therapeutic Vehicles-Gold Nanoparticles
Gold nanoparticles are a kind of particles with a structure composed of gold atoms at the nanometer level. Due to the size of the nanometer, it has unique properties in chemistry, physics, optics, electronics, etc., and has been widely used in the field of nanomedicine. As a kind of inorganic nanoparticles, gold nanoparticles are used for imaging and targeted drug delivery due to their unique advantages.
Classification and Application of Gold Nanoparticles
Gold nanomaterials have become a new hot spot in the research and development of anti-tumor drugs in recent years due to their good biocompatibility and local surface plasmon resonance that can be adjusted in the near-infrared region. Common gold nanostructures mainly include gold nanorods, gold nanostars, and gold nanospheres. One can obtain gold nanoparticles with a diameter of 1 to 150 nm and different shapes and characteristics by adjusting the synthesis conditions, and apply them to the construction of drug delivery systems. Spherical gold nanoparticles are the most common and the earliest researched type of gold nanoparticles. Most of them have a diameter of 1-100 nm and show different colors with the change of particle size. A large number of studies have confirmed that spherical gold nanoparticles have strong binding ability to thiols, disulfides and amines, and can quickly and stably bind nucleic acids, polypeptides, proteins, small molecule compounds and other substances through such groups. The binding maintains the stability and biological activity of the loaded molecules in internal and external environments.
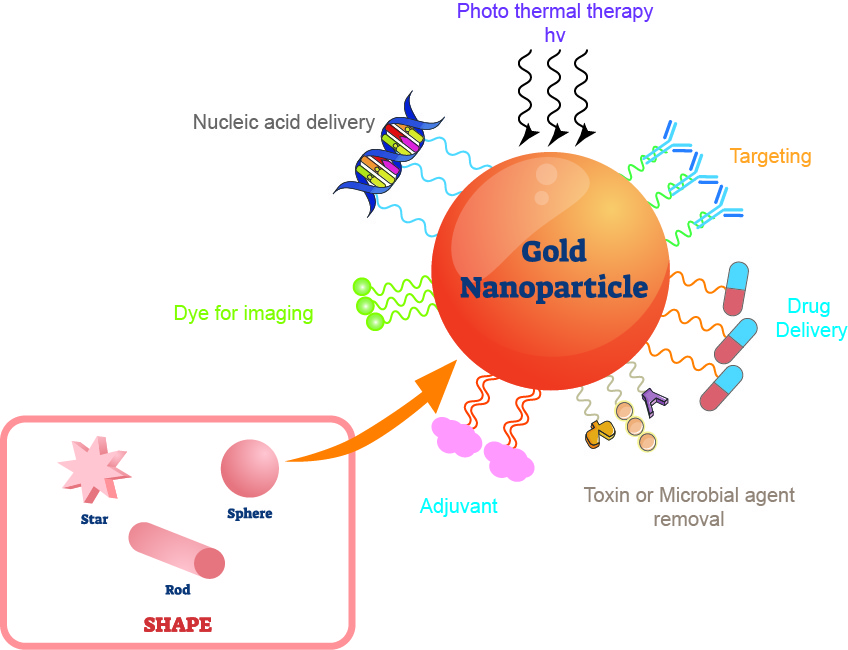
Figure 1. The application of gold nanoparticles.
In the follow-up more in-depth research on gold nanoparticles, it is found that changes in the shape of gold nanoparticles often give them new characteristics. Certain shapes of gold nanoparticles can often generate heat under the irradiation of a specific wavelength of laser light, which can kill tumor cells while promoting the breaking of chemical bonds on the surface to release anti-tumor drugs. Gold nanorods and hollow gold nanoshells are gold nanoparticles with rod-like and hollow spherical structures, respectively, and possess a light-to-heat conversion efficiency that spherical gold nanoparticles (solid structures) do not have. With the change of the aspect ratio of the gold nanorods, the surface plasmon resonance wavelength will also change at 550~1 550 nm, and it has a high surface electric field enhancement effect, high optical absorption, high scattering cross section, and continuous adjustable light-to-heat conversion efficiency from 50% to 100%. The light-to-heat conversion efficiency of the hollow gold nanoshell is weaker than that of the gold nanoshell, but the hollow spherical structure can lead to a higher specific surface area, and thus obtain the advantages of high loading and easy surface functionalization. In recent years of research, gold nanocages have gradually entered people’s field of vision. While they have high efficiency of light-to-heat conversion, their hollow and porous structure also gives them better drug-carrying efficiency than other gold nanoparticles. The discovery of enhanced permeability and retention effect (EPR) is the main reason why early nano-drug delivery systems including nano-gold have received widespread attention in the field of anti-tumor research. This effect can promote the accumulation of nano-sized substances in solid tumor tissues, and is often used for tumor-targeted delivery of nano-particles. While enhancing the curative effect, it reduces adverse reactions to healthy tissues and organs. In recent studies, people often modify the surface of nano-gold materials with tumor-targeting functional molecules to further enhance their specificity to tumor tissues on the basis of the EPR effect. With the gradual deepening of research on the transformation of gold nanoparticles, the drug loading of gold nanoparticles continues to increase, and redox and pH sensitivity design have also been introduced into the gold nanoparticle drug delivery system. Researchers use the acidic microenvironment and high content of redox substances in tumor tissues to improve the targeted release efficiency of the modified gold nanoparticle delivery system. Advances in these technologies continue to push anti-cancer drugs based on gold nanoparticles to the clinic.
In recent years, a variety of gold nanoparticles have been used in imaging and photothermal therapy. Photothermal therapy is a treatment that uses the photothermal effect of photothermal conversion materials to convert external light energy into heat energy, which causes the local temperature of tumor cell nuclear tissue to rise rapidly, resulting in irreversible damage and denaturation of DNA and protein, thereby inducing tumor cell death method. This therapy has been used since the beginning of the twentieth century. In traditional hyperthermia, the use of laser irradiation to raise the temperature of the local area of the tumor to 42–45 °C can destroy cancer cells and tissues. Simple gold nanospheres (AuNPs) and gold nanorods (AuNRs) are generally difficult to load drugs, and their surface needs to be modified when applied to tumor diagnosis and treatment. In 2015, Chen et al. designed a pH-responsive drug delivery system based on AuNPs. They combined the pH-responsive anti-tumor drug AD-Hyd-DOX and tumor targeting peptide AD-PEG8-GRGDS on AuNP@CD through self-assembly and simultaneously modified them to obtain a multifunctional AuNP@CD-AD-DOX/RGD nanocomposite material . This composite nanoparticle can increase the intake of tumor cells by RGD, and release the loaded drug under the acidic conditions of the tumor microenvironment, which can significantly inhibit the growth of tumor cells. In 2017, Sun et al. used gold nanoparticles modified with heat-sensitive peptide ELP to self-assemble under high temperature conditions to form a gold nanosphere assembly, which exhibits strong near-infrared light absorption and high photothermal effect. These characteristics make it possible to achieve The dual functions of tomographic imaging and photothermal therapy of tumors are realized. Keul et al. prepared a series of gold nanorods (AuNRs) with different aspect ratios by seed growth. As their aspect ratios increase, the local surface plasmon resonance peaks (LSPR) of AuNRs can gradually redshift from the visible light region to Near infrared light area. The unique optical properties of AuNRs make it more potential than AuNPs in tumor hyperthermia.
References:
1.Chen Z, et al.; Sandwich-type Au-PEI/DNA/PEI-Dexa nanocomplex for nucleus-targeted gene delivery in vitro and in vivo. ACS Appl Mater Interfaces. 2014; 6:14196-206.
2.Sun L, et al.; Functional gold nanoparticle-peptide complexes as cell-targeting agents. Langmuir. 2008; 24:10293-7 .
3.Heidrun A. Keul, et al.; Structural Evolution of Gold Nanorods during Controlled Secondary Growth. Langmuir. 2007, 23, 20, 10307–10315.
4.Mohamed Kodiha, et al.; Off to the Organelles - Killing Cancer Cells with Targeted Gold Nanoparticles. Theranostics. 2015; 5(4):357-370.


