Oral Delivery
 CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. We also have developed mature technology platforms for drug delivery, such as inorganic nanomaterials, biomacromolecules, polymeric and lipid system. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy. We are dedicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial researchers and assay developers all around the world. One of our focus areas is oral drug delivery which is defined as the administration of pharmaceutically active agents taken through the mouth to obtain local or systemic effects.
CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. We also have developed mature technology platforms for drug delivery, such as inorganic nanomaterials, biomacromolecules, polymeric and lipid system. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy. We are dedicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial researchers and assay developers all around the world. One of our focus areas is oral drug delivery which is defined as the administration of pharmaceutically active agents taken through the mouth to obtain local or systemic effects.
Introduction to Oral Delivery
As the most preferred drug administration route, oral drug delivery is convenient, cost-effective and easily accepted by patients. However, there are still some challenges for this route, such as the aqueous solubility, membrane permeability and chemical and enzymatic stability of drugs.
As a noninvasive method, patients are usually familiar with taking orally delivered drugs, which could avoid pain and discomfort associated with injections as well as eliminating contaminations. In addition, due to their safety, efficacy, economic and consumer compliance advantages, oral dosage forms take more majority of the drug delivery market than other routes of delivery. It is estimated that about 80% of all medications used today is through the oral route, and people still prefer the tablets, capsules, and granules as their No.1 choice for the dosage form. As a result, it is vital to improve its safety and efficacy for oral drug delivery. However, the barriers to oral drug delivery (such as the solubility and dissolution, absorption, presystemic metabolism, and excretion) cannot be ignored too. Only the portion of drugs that overcome the barriers like the blood-brain barrier and then reach their targets, such as the central nervous system or cancer cells can be considered bioavailable. So, efforts must be made to overcoming these barriers to improve the performance of orally delivered drugs.
Generally, for all oral drugs, they can only be absorbed after being dissolved in the aqueous environment of the gastrointestinal (GI) tract. This easy step is actually the most challenge part for developing a new oral drug, such as developing drugs with lipophilic molecules that can hardly be soluble in aqueous solutions. For example, the transit time for undissolved drug solids in the upper GI tract- stomach and small intestine, is usually about 2 to 6 hours. We must make sure the dissolution rates are sufficiently high so that there is enough time to dissolve these solids for absorption.
The first step is to dissolve in the gastric fluid. Then, drug components should keep soluble in the entire GI tract. In the view of the pH of the GI tract differs dramatically (pH= 1.5 in the stomach, pH= 8 in the lower small intestine and colon), this goal is not easily achieved. Because pH would alter the solubility of many drugs, when designing solubility and dissolution experiments, we must take this factor into consideration.
After being dissolved, drugs will be absorbed by the means of different transcellular and paracellular pathways in the intestinal tract. Drugs being transported between cells is absorbed through the paracellular pathway by passive diffusion or carrier-mediated transport, while drugs being transported across the cells through the apical and basolateral membranes is absorbed through transcellular pathway mainly by passive diffusion. Due to the large absorption area (around 100 m2, which is equal to a quarter area of a basketball court), the main absorption site of drugs is the small intestine. Although the absorption of drugs in the colon varies with the undigested materials, bacteria, and variable water content, GI transit still plays a key role in the absorption of drugs.
When drugs are absorbed into intestinal cells, some of them would experience the first-pass metabolism by different kinds of intestinal metabolic enzymes. For drugs escaped from the above procedure, their bioavailability is still not guaranteed because there is an additional first-pass metabolism by the liver. All these metabolites would be directly or indirectly excreted back to the lumen or be excreted via the bile. In addition, they may function in the blood, followed by the elimination via the kidneys.
For the last few years, numerous studies have been done to improve the performance of oral delivery, especially for the improvement of oral delivery of therapeutic peptides and proteins. To enhance drug and vaccine oral delivery, researchers have developed various strategies. They find that drugs associated with colloidal carriers, for example, polymeric nanoparticles could improve their oral bioavailability (Figure 1).
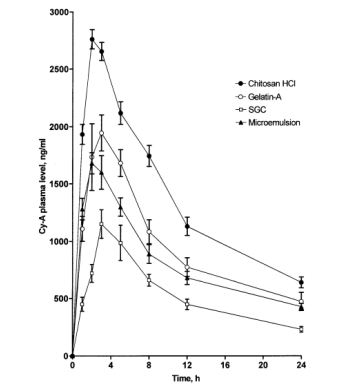
Figure 1. Mean plasma level of Cy-A following oral administration to beagle dogs. Particle size reduction of Cy-A to positively charged nanoparticles using cationic polymers such as chitosan HCl or gelatin-A improved its absorption rate and overall bioavailability. (El-Shabouri, M.. (2002). Positively charged nanoparticles for improving the oral bioavailability of cyclosporin-A. International Journal of Pharmaceutics, 249(1-2), 101–108.)
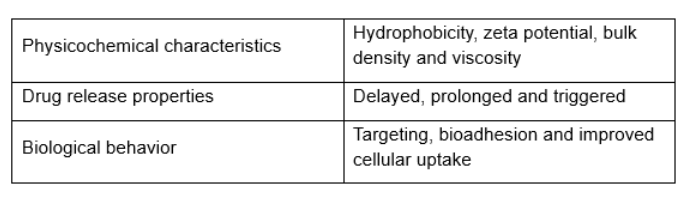
Polymeric nanoparticles are with pharmaceutical advantages: 1) Comparing to other colloidal carriers like liposomes, they are more stable in the GI tract and can prevent encapsulated drugs damaging from the GI environment. 2) There are various polymeric materials which enrich the possibility of modulation of physicochemical characteristics, drug release properties and biological behavior (Table 1). 3) After adsorbing or chemical grafting of some molecules (e.g. PEG and poloxamers) and bioactive molecules (e.g. lectins and invasins), the surface of the particle can be modified. In addition, the advantage of their submicron size and large specific surface area makes polymetric nanoparticles easier to absorb than larger carriers. Many studies have demonstrated that nanoencapsulation of peptides and protein colloidal particles protects them against the adverse environment of the GI tract, and enhances their transmucosal transport.
Table 1. Modifiable properties of nanoparticles.
Our Featured Services
CD Bioparticles is specialized in the development of drug delivery systems and customizing nanoparticles for drug delivery utilizing our core technologies. With our high-quality products and services, the efficacy of your drug delivery can be tremendously improved.
We offer custom synthesis of polymer microspheres and nanoparticles. Clients may select the material type, particle size, size distribution, color dye, fluorescent dye, and/or surface functional groups such as carboxyl or amine groups. We also encapsulate proteins, steroids, ligands, nucleic acids, and other drug molecules. Additionally, the surface coating of microspheres or nanoparticles with ligands, oligonucleotides, and other agents are available for clients to choose.
References:
1. Des Rieux, A., Fievez, V., Garinot, M., Schneider, Y.-J., & Préat, V. (2006). Nanoparticles as potential oral delivery systems of proteins and vaccines: A mechanistic approach. Journal of Controlled Release, 116(1), 1–27.
2. Gupta, U., & Perumal, O. (2014). Dendrimers and its biomedical applications. Natural and Synthetic Biomedical Polymers, 243–257.
3. Zhu, L., Lu, L., Wang, S., Wu, J., Shi, J., Yan, T., Liu, Z. (2017). Oral Absorption Basics. Developing Solid Oral Dosage Forms, 297–329.


 CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. We also have developed mature technology platforms for drug delivery, such as inorganic nanomaterials, biomacromolecules, polymeric and lipid system. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy. We are dedicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial researchers and assay developers all around the world. One of our focus areas is oral drug delivery which is defined as the administration of pharmaceutically active agents taken through the mouth to obtain local or systemic effects.
CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. We also have developed mature technology platforms for drug delivery, such as inorganic nanomaterials, biomacromolecules, polymeric and lipid system. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy. We are dedicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial researchers and assay developers all around the world. One of our focus areas is oral drug delivery which is defined as the administration of pharmaceutically active agents taken through the mouth to obtain local or systemic effects.

