Passive Drug Targeting by Modifying Particle Size, Shape and Surface Characteristics
Passive targeting of nanoparticles (NPs) takes advantage of the abnormal tumor physiology and structure that results in the EPR effect. Particles’ circulation time, targeting, and the ability to overcome biological barriers can be modified by changing its size, particle shape, and surface characteristics. CD Bioparticles offers custom services for particle synthesis and modification used in passive drug delivery system.
Introduction to Influence of Particle Size, Shape and Surface Characteristics on Passive Targeting
The distribution and accumulation of NPs in tumors are also multifaceted and are affected by the biological and physicochemical properties of each material. In passive targeting, macromolecules including NPs accumulate preferentially in the tumor tissues as a result of the enhanced permeability and retention (EPR) phenomenon. Modifying physiological properties such as particle size, shape and surface characteristics of particles will have a deep effect on their performance.
Passive targeting is largely possible through diffusion-mediated transport, which makes size a critically important factor. The size of the nanomaterial influences the kinetics and extent of tumor accumulation. Conventionally, the optimal size range of a particle is 40-200 nm, which will ensure longer circulation time, increased accumulation within the tumor mass and lower renal clearance. It has been noted that a nanoparticle must be at least 10 nm in diameter to avoid clearance by first pass renal filtration. Furthermore, the distribution of smaller particles is usually more homogenous.
Like particle size, particle shape the curvature also plays an important role in cellular uptake within the tumor. NPs may exhibit an extensive range of geometries—from spherical to tubular, through centric, eccentric and star like. While spherical NPs are good candidates for drug delivery, anisotropic structures can sometimes provide higher efficiencies in drug deliver (due to a more favorable configuration with the cell), although the sharp edges and corners may induce injuries to blood vessels. NPs may be hollow, dense, nanostructured, or in core-shell (eventually with multiple cores) structures, enhancing the NP load capacity and specific targeting ability.
One of the studies demonstrated that the cylindrical particles had a greater uptake into the cells. Also, the aspect ratio of the nanoparticles plays a very important role in determining their uptake. Another study showed that particles with 150 nm diameter and 450 nm heights were taken into the cells approximately four times faster than symmetrical particles with an aspect ratio of 200. A general statement is that the rigid, spherical particles, which are particularly 100-200 nm in size, have the greatest propensity for prolonged circulation because they are large enough to avoid any liver uptake, but at the same time, are optimal in size to avoid filtration in the spleen.
Surface characteristics also effect much in determining the extent of internalization of these NPs into cells. Relatively, the surface can be modified by the polymer composition, thus governing an extra amount of hydrophobicity or hydrophilicity to these particles. The presence of surface charge can alter the opsonisation profile of the material, its recognition by cells in the organs of the MPS and its overall plasma circulation profile. Negative surface charges can either increase, decrease or have no impact on the blood clearance of NPs, but positive charges are generally recognized as having a negative effect on the plasma exposure to the nanomaterial. For example, coating the nanoparticle surface with a hydrophilic polymer (like PEG) protect the nano-systems from opsonization and subsequent clearance by the reticuloendothelial system (RES). In addition, increasing the molecular weight of hydrophilic polymer chains will also increase the circulation time of these NPs.
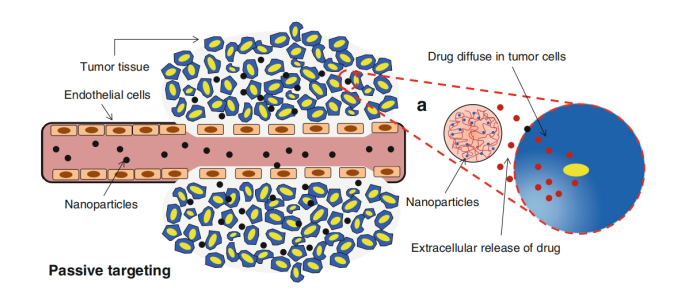
Figure 1. By the enhanced permeability and retention effect, NPs passively diffuse through the leaky vasculature and accumulate in tumor tissues. (Pawar, Prasad V., et al. Focal Controlled Drug Delivery, Springer US: Boston, MA, 2014, 61-91.)
Our Modifying Particle Featured Services
Since passive targeting can be regulated by modifying the size, shape or in some cases, the surface dimensions of these nanoparticles, our experienced scientists are proficient in synthesizing nanocarriers with special required properties as follows.
|
Size
|
10-400 nm
|
|
Shape
|
Spheres, cubes, stars, bipyramids, clusters, hollows, cages, shuttles, and so on.
|
|
Surface coating
|
polymers (PEG, PLGA, PLA, PEI, chitosan, dextran, copolymers), lipid, silane, and so on.
|
Quotations and Ordering

References:
1. Wakaskar RR. Passive and active targeting in tumor microenvironment. Int. J. Drug Dev. Res, 2017, 9, 37-41.
2. Sykes, Edward A., et al. Investigating the impact of nanoparticle size on active and passive tumor targeting efficiency. ACS Nano, 2014, 8(6), 5696-5706.
3. Steichen, S. D., Caldorera-Moore, M., Peppas, N. A. A review of current nanoparticle and targeting moieties for the delivery of cancer therapeutics. European Journal of Pharmaceutical Sciences, 2013, 48(3), 416-427.



