Pharmacodynamics and Pharmacokinetics Test
With advanced technical equipment and years of experience in bio-pharmaceuticals, CD Bioparticles provides customer-oriented services to measure the pharmacodynamics and pharmacokinetics of drugs delivered by bioparticles.
Pharmacodynamics and Pharmacokinetics Introduction
Pharmacokinetics is currently defined as the study of the time course of drug absorption, distribution, metabolism, and excretion. Primary goals of pharmacokinetics include enhancing efficacy and decreasing toxicity of drug therapy. These parameters have enabled clinicians to apply pharmacokinetic principles to actual patient situations. Pharmacodynamics refers to the relationship between drug concentration at the site of action and the resulting effect, including the time course and intensity of therapeutic and adverse effects. Pharmacodynamics places a particular emphasis on dose–response relationships, it provides an important parameter for the clinical application of drugs.
Pharmacodynamics and Pharmacokinetics Test Parameters
The most commonly measured pharmacodynamic and pharmacokinetic metrics are listed below.
Pharmacodynamics:
-
Liberation – the process of release of a drug from the pharmaceutical formulation.
-
Absorption – the process of a substance entering the blood circulation.
-
Distribution – the dispersion or dissemination of substances throughout the fluids and tissues of the body.
-
Metabolism (or biotransformation, or inactivation) –the foreign substance is recognized and irreversibly transform from parent compounds into daughter metabolites.
-
Excretion – the removal of the substances from the body. In rare cases, some drugs irreversibly accumulate in body tissue.
Pharmacokinetic:
-
Dose, amount of drug administered.
-
Dosing interval, time between drug dose administrations.
-
Cmax, the peak plasma concentration of a drug after administration.
-
Tmax, time to reach Cmax.
-
Cmin, the lowest (trough) concentration that a drug reaches before the next dose is administered.
-
Volume of distribution, the apparent volume in which a drug is distributed (i.e., the parameter relating drug concentration in plasma to drug amount in the body).
-
Concentration, amount of drug in a given volume of plasma.
-
Elimination half-life, the time required for the concentration of the drug to reach half of its original value.
-
Elimination rate constant, the rate at which a drug is removed from the body.
-
Infusion rate, rate of infusion required to balance elimination.
-
Area under the curve, the integral of the concentration-time curve (after a single dose or in steady state).
-
Clearance, the volume of plasma cleared of the drug per unit time.
-
Bioavailability, the systemically available fraction of a drug.
-
Fluctuation, peak trough fluctuation within one dosing interval at steady state.
The Methods of Pharmacodynamics and Pharmacokinetics Test
The handling of a drug by the body can be very complex, as several processes (such as absorption, distribution, metabolism, and elimination) work simultaneously to alter drug concentration in tissues and fluids. Simplifications of body processes are necessary to predict a drug’s behavior in the body. One way to make these simplifications is to apply mathematical principles to the various processes. Thus, a basic type of body model, compartmental model, has been used in pharmacokinetics. Compartmental models are categorized by the number of compartments needed to describe the drug’s behavior in the body. There are one-compartment, two-compartments, and multicompartment models. The compartments do not represent a specific tissue or fluid but may represent a group of similar tissues or fluids. These models can be used to predict the time course of drug concentrations in the body.
Microscale Thermophoresis
Microscale thermophoresis is a bioanalytical method that can be used to quantify how the biological matrix/liquid affects the affinity of a drug to its target. A concentration-time profile will be constructed by employing chemical techniques to measure the concentration of drugs in biological matrix, most often plasma.
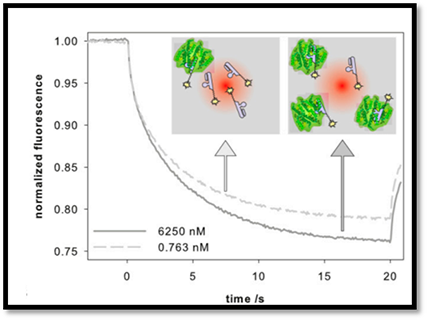
Figure 1. Microscale thermophoresis (MST) curves.( Peggy R., et al. Int. J. Mol. Sci. 2017, 18, 2484)
Mass Spectrometry
Pharmacokinetics is often studied using mass spectrometry because of the complex nature of the matrix (often plasma or urine) and the need for high sensitivity to observe concentrations after a low dose and a long time period. Here, we use the most common instrumentation LC-MS with a triple quadrupole mass spectrometer for pharmacokinetics study.
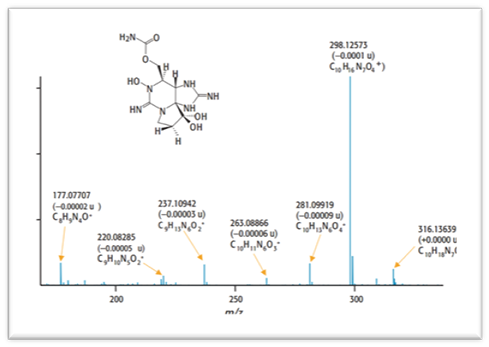
Figure 2. Ion spectrum of FT-ICR product. (Dietrich A., et al. 2004, Spectroscopy 19(6):16-24.)
CD Bioparticles offers a full set of service for pharmacodynamics and pharmacokinetics study. For more detailed information, please feel free to contact us or directly send us an inquiry.
Quotations and Ordering

References:
1. Lees P, et al. Principles of pharmacodynamics and their applications in veterinary pharmacology. J. Vet. Pharmacol. Ther. 2004, 27 (6): 397–414.
2. Ruiz G. A., et al. Pharmacokinetics in drug discovery. Journal of Pharmaceutical Sciences. 2008, 97 (2): 654–90.
3. AGAH working group PHARMACOKINETICS (2004-02-16). Collection of terms, symbols, equations, and explanations of common pharmacokinetic and pharmacodynamic parameters and some statistical functions. AGAH. Retrieved 2011-04-04.
4. Baaske P, et al. Optical thermophoresis for quantifying the buffer dependence of aptamer binding. Angewandte Chemie. 2010, 49 (12): 2238–41.
5. Hsieh Y, Korfmacher WA. Increasing speed and throughput when using HPLC-MS/MS systems for drug metabolism and pharmacokinetic screening. Current Drug Metabolism. 2006, 7 (5): 479–89.




