mRNA-LNP Vaccine Laboratory Process Development Assay
The mRNA molecule is well known to the public due to the COVID-19, and the mRNA vaccine made from it has the characteristics of blocking the reaction speed against pathogen mutation, simple production process, and scale-up. Detection and validation methods are the basis for quality control of mRNA-LNP vaccines; for the characteristics of mRNA, liposomes and preparations, as well as related impurities and other key quality attributes of mRNA vaccines, R&D companies should establish detection methods to ensure the safety and efficacy of mRNA-LNP vaccines.
Characterizations of mRNA-LNP
mRNA-LNP morphology can be identified using cryo-TEM. The polydispersity index PDI, zeta potential, mRNA identification and concentration, and mRNA encapsulation efficiency (%) will also be determined.
An example of the quality of mRNA-LNP was characterized. (Refer to Figure 1)
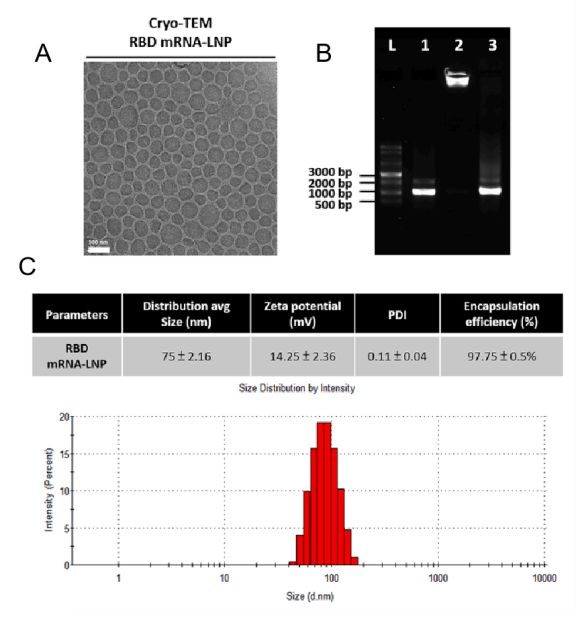 Figure 1. Characterization of mRNA-LNP.[1]
Figure 1. Characterization of mRNA-LNP.[1]
Cellular uptake tests can be used to study endocytosis pathway. Cy5-mRNA, a model mRNA, can be utilized to prepare Cy5-mRNA-LNP. Different inhibitors were co-incubated with cell, then the inhibitors will be replaced with Cy5-mRNA-LNP and kept for certain hours. Finally, cells were collected and analyzed by flow cytometry. The commonly used inhibitors include:
1. Filipin to block fovea-mediated endocytosis.
2. Cytochalasin-D to block actin-associated cytosolic effluent.
3. Wortmannin to block phosphatidylinositol 3 kinase-associated cytosolic effluent.
4. Chloropromazine to block reticulin-mediated endocytosis.
Cy5 staining allows real-time tracking of mRNA cellular uptake, cellular internalization, and lysosomal escape at different times, which helps to study the mechanism behind mRNA delivery. It can also further optimize the formulation of LNP by exploring the release of mRNA in vivo. (Refer to Figure 2)
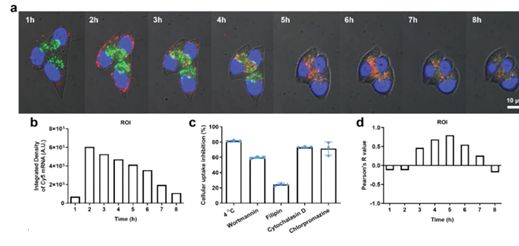 Figure 2. Cellular uptake, intracellular trafficking, and lysosomal escape of LNPs.[2]
Figure 2. Cellular uptake, intracellular trafficking, and lysosomal escape of LNPs.[2]
- mRNA Transfection Efficiency Study
DC2.4 cells will be selected as the model cells for the contagion experiment. GFP-mRNA-LNP was contagious to the cells and the intracellular GFP signals were observed under the fluorescence microscope. And the transfection efficiency was analyzed by flow cytometry.
CD Bioparticals can assist customers with immunostaining studies, which not only examine the efficiency of mRNA transmission, but also help to identify specific cell types that are transfected to a particular type of organ. (Refer to Figure 3)
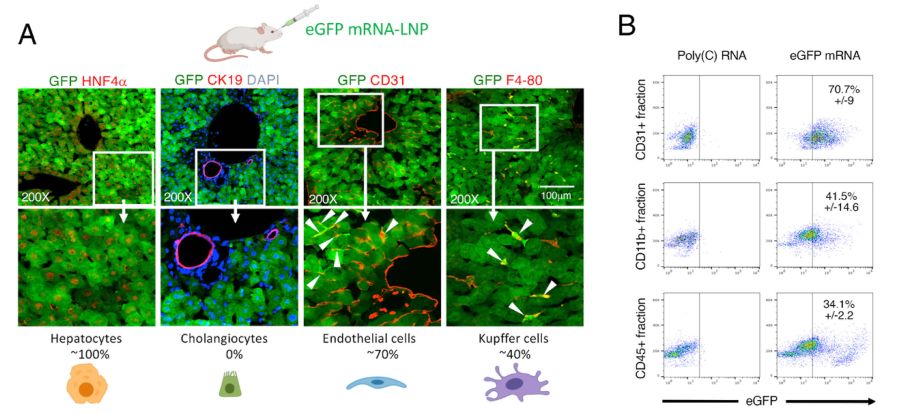 Figure 3. Identification of transfected liver cell types 5 h after IV eGFP mRNA-LNP injection.[3]
Figure 3. Identification of transfected liver cell types 5 h after IV eGFP mRNA-LNP injection.[3]
To visualize LNP distribution observation in organisms, fluorescent dyes (such as DiD and DiO) and luciferase will be used for labeling. Typically, we used luciferase mRNA (Luc-mRNA) as a model mRNA for tracking the in vivo distribution of the expressed protein. Following intravenous (IV) injection of Luc-mRNA-LNP, heart, liver, spleen, lung, kidney, and lymph node tissues will bd isolated. The tissues will be visualized using the IVIS Spectral Live Image System. This will help to visualize the study of organ distribution and duration of protein activity production. (Refer to Figure 4)
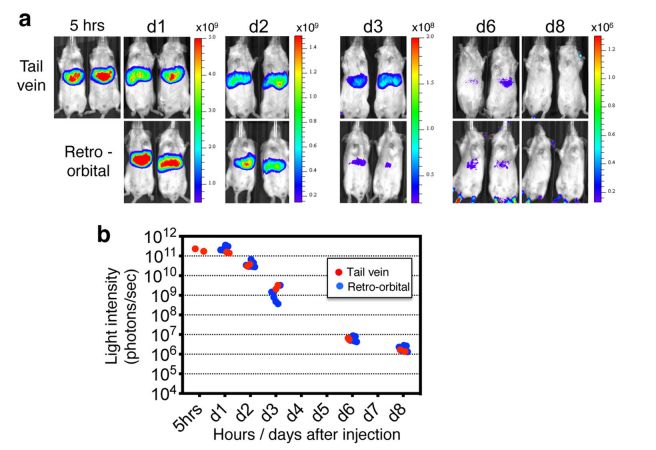 Figure 4. Visualization of the expression by an in vivo imaging system.[4]
Figure 4. Visualization of the expression by an in vivo imaging system.[4]
- Histopathological Studies
A experimental animal model (e.g., mice, rabbits, pigs, etc.) will be established with cells expressing the relevant antigens and the experimental animals will be immunized intravenously every few days. The experimental animals will be administered and tested daily for body weight and tumor volume. Subsequently, the experimental animals will be executed to isolate the heart, liver, spleen, lung and kidney tissues for weighing. Spleen, lymph nodes and tumors will be collected to determine the maturation of DCs and the number of CTLs respectively. (Refer to Figure 5)
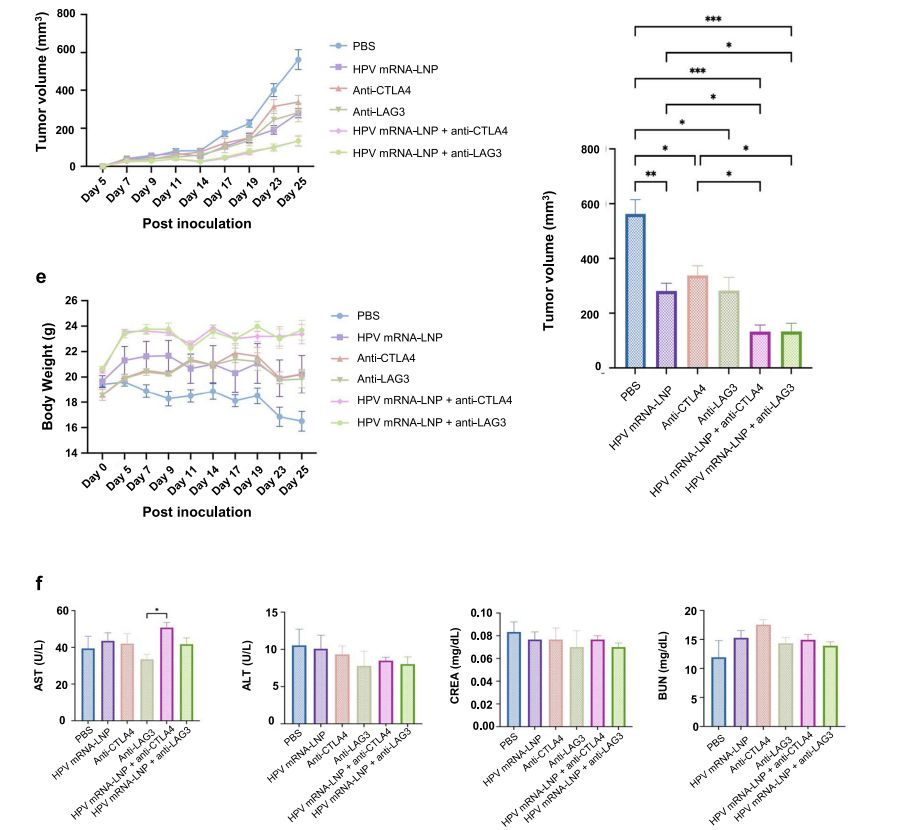 Figure 5. Determination of body weight and tumor volume in mice.[4]
Figure 5. Determination of body weight and tumor volume in mice.[4]
Sections of organs are taken and stained for pathologic observation. The commonly used pathologic staining methods include:
1. H&E staining method. (Using two dyes, Hematoxylin and Eosin, the former stains the nucleus and some acidic cytoplasm, the latter stains the cytoplasm and collagen fibers, and the tissue sections stain to show blue and pink morphology).
2. PAS staining method. (Using two kinds of dyes, Periodicacid and Schiff'sreagent, the tissue sections are stained with purplish-red color).
3. Masson staining method. (Mainly used to show connective tissues such as collagen fibers, skeletal muscle fibers, etc. Using two dyes, Anilineblue and Picro-fuschin, the tissue sections show red and blue coloration after staining).
For example, H&E stained for pathological observation. Briefly, the tissue section is stained with Gill's Hematoxylin, blued with ammonia, washed in ethanol, stained with 0.25% Eosin Y. Then the stained section will be characterized under bright-field microscope. Recovery of organ function can be detected by observation of immunologic tissue. (Refer to Figure 6)
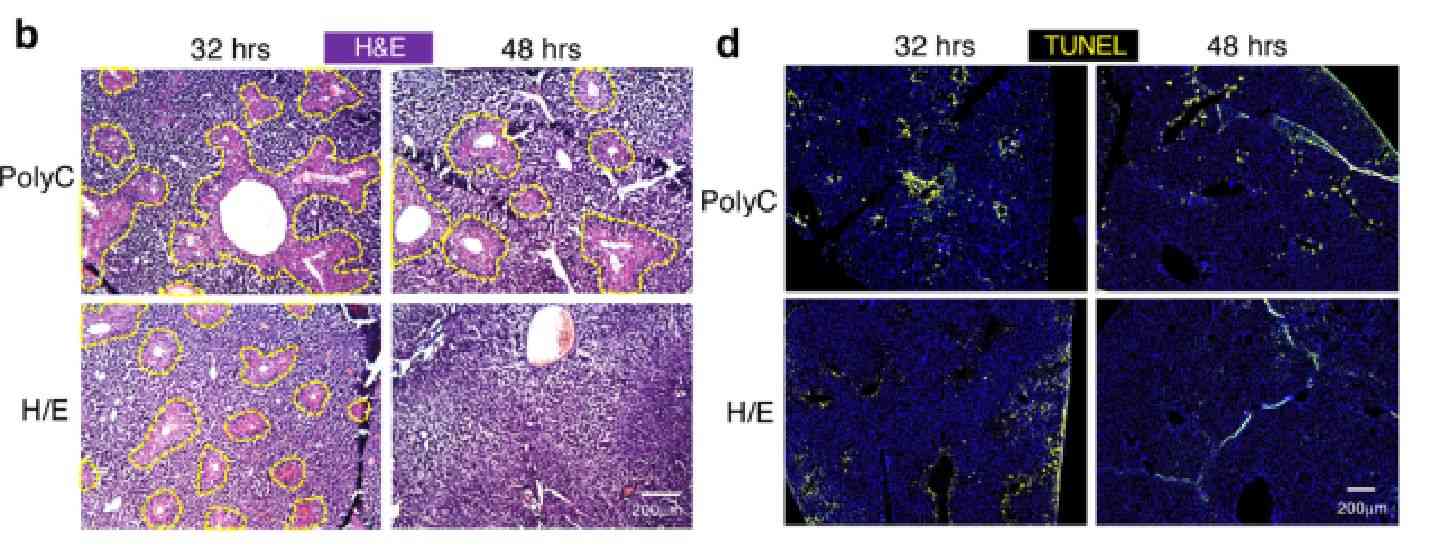 Figure 6. Tissue section staining studies.[4]
Figure 6. Tissue section staining studies.[4]
- Biochemical Indicators Testing
Blood can be taken from the eyeballs to determine alanine aminotransferase (ALT), aspartate aminotransferase (AST), total protein (TP), uric acid (UA), urinary anhydride (UREAL) and other biochemical indexes to detect toxicity of mRNA-LNP to the liver and kidney. (Refer to Figure 7)
The assay was performed using the kit according to manufacturer's method. Briefly, 10 μL of serum was mixed with the provided reagent mixture at 37℃ and readings were measured at 5 nm every 1 minute for 3 minutes using a multi-mode enzyme marker.
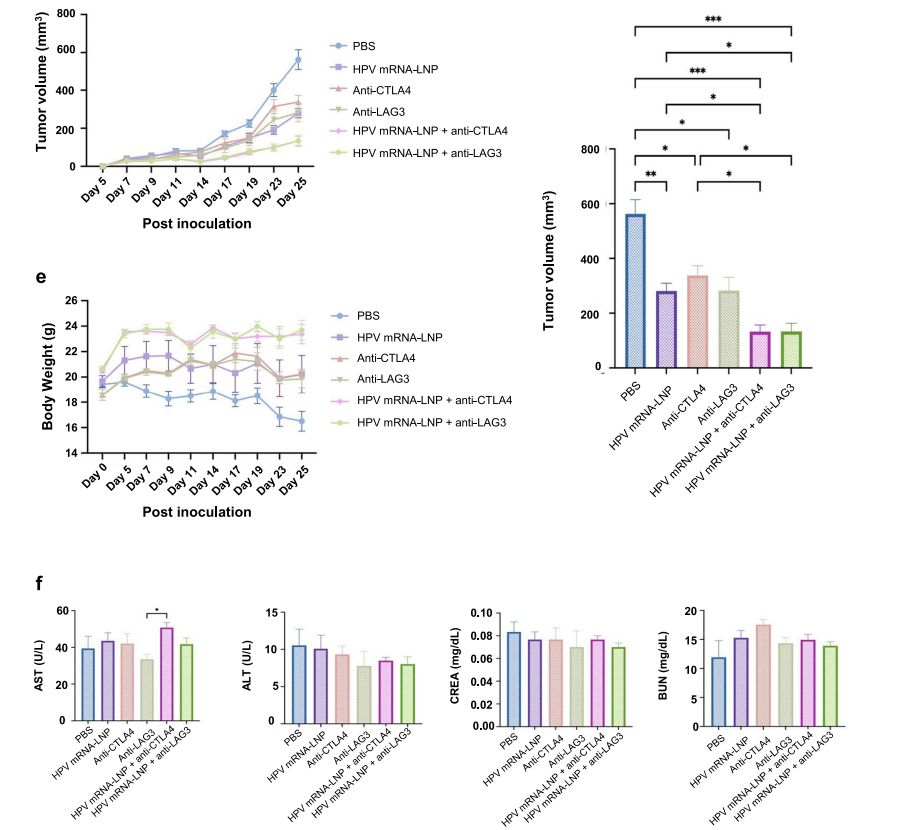 Figure 7. Determination of parameters related to mRNA-LNP administration therapy.[5]
Figure 7. Determination of parameters related to mRNA-LNP administration therapy.[5]
- Detection of antigen-specific T cells
A hormonal mouse model is established and injected with a therapeutic vaccine. Mice will be executed and spleen, lymph and tumor were collected for detection of antigen-presenting and antigen-specific T cells. Cells are labeled with appropriate markers and analyzed and detected by flow cytometry.
References
- Hsu FF, et al.; An efficient approach for SARS-CoV-2 monoclonal antibody production via modified mRNA-LNP immunization. International Journal of Pharmaceutics. 2022, 627, 122256.
- Everton E, et al.; Transient yet robust expression of proteins in the mouse liver via intravenous injection of lipid nanoparticle-encapsulated nucleoside-modified mRNA. Bio-protocol journal. 2021, 11(19): e4184.
- Zhang R, et al.; Esterase-labile quaternium lipidoid enabling improved mRNA-LNP stability and spleen-selective mRNA transfection. Advanced materials. 2023, 3, 614.
- Rizvi F, et al.; Murine liver repair via transient activation of regenerative pathways in hepatocytes using lipid nanoparticle-complexed nucleoside-modified mRNA. Natrue Communications. 2021, 12, 613.
- Diu K, et al.; mRNA-LNP vaccination-based immunotherapy augments CD8+ T cell responses against HPV-positive oropharyngeal cancer. npj Vaccines. 2023, 144, 0733.


 Figure 1. Characterization of mRNA-LNP.[1]
Figure 1. Characterization of mRNA-LNP.[1] Figure 2. Cellular uptake, intracellular trafficking, and lysosomal escape of LNPs.[2]
Figure 2. Cellular uptake, intracellular trafficking, and lysosomal escape of LNPs.[2] Figure 3. Identification of transfected liver cell types 5 h after IV eGFP mRNA-LNP injection.[3]
Figure 3. Identification of transfected liver cell types 5 h after IV eGFP mRNA-LNP injection.[3] Figure 4. Visualization of the expression by an in vivo imaging system.[4]
Figure 4. Visualization of the expression by an in vivo imaging system.[4] Figure 5. Determination of body weight and tumor volume in mice.[4]
Figure 5. Determination of body weight and tumor volume in mice.[4] Figure 6. Tissue section staining studies.[4]
Figure 6. Tissue section staining studies.[4] Figure 7. Determination of parameters related to mRNA-LNP administration therapy.[5]
Figure 7. Determination of parameters related to mRNA-LNP administration therapy.[5]