Solid Lipid Nanoparticle Production
CD Bioparticles provides custom services for solid lipid nanoparticle production to prolong drug circulation time. Based on our deep understanding of solid lipid nanoparticle, our scientist team can provide professional solutions for solid lipid nanoparticle preparation and characterization.
Introduction to Solid Lipid Nanoparticles
Solid lipid nanoparticles typically contain a hydrophobic solid matrix core with one layer of a phospholipid coating (Figure 1), and it is typically spherical with an average diameter between 10 and 1000 nanometers. Solid lipid nanoparticles are prepared by using solid lipids (e.g., purified triglycerides, fatty acids, steroids, or waxes) that can solubilize lipophilic molecules, emulsifiers that stabilize the core, and water. The hydrophobic tail regions of the phospholipid are embedded into the core matrix resulting in the core exclusively possesses a hydrophobic nature for drug encapsulation. Solid lipid nanoparticles are a new generation of colloidal drug carrier systems and consist of surfactant-stabilized lipids that are solid both at room and body temperatures, and solid lipids are melted by heating to 5–10°C above the melting temperature, then the drug is dissolved, dispersed, or solubilized in the hot melted lipids. The active drug may incorporate into the solid lipid nanoparticles in three different models including the homogenous matrix model, the drug-enriched shell model, and the drug-enriched core model.
In general, solid lipid nanoparticles have a higher entrapment efficiency for hydrophobic drugs in the core compared with conventional liposomes, and they are are also considered to well tolerated due to their composition from physiologically similar lipids. In addition to its prolonged drug release feature, solid lipid nanoparticles also have advantages with its low toxicity, biodegradable composition, and high stability against aggregation or coalescence, could be easy scale-up with low cost of production. The solid lipid nanoparticles are suitable for intravenous administration and can be successfully dispersed in aqueous or aqueous-surfactant solutions.
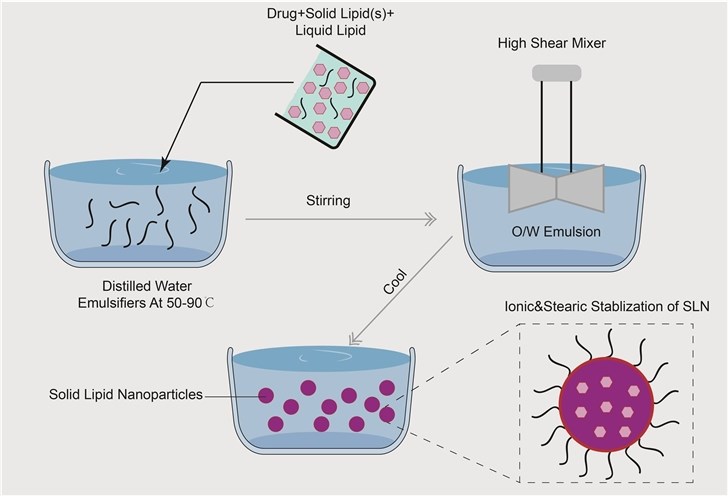 Figure 1. Illustration of solid lipid nanoparticles and drug loading.
Figure 1. Illustration of solid lipid nanoparticles and drug loading.
Solid Lipid Nanoparticles Applications
Due to its lipid structured matrix, solid lipid nanoparticles enhance drug encapsulation and stability which made them popular in nano pharmaceutical research field and other applications. Solid lipid nanoparticles have a remarkably wide range of application and have shown greatly to control the skin penetration of several actives, delivery of food and drugs, cosmetics and other applications. Solid lipid nanoparticles have recently materialized as a novel approach to oral and parenteral drug delivery systems for poorly water-solubility of drugs. Solid lipid nanoparticles could also be functioned with other targeting strategies to improve its performance in drug delivery.
Our Solid Lipid Featured Services
-
Liposome formulation design: we customize liposomes design based on our clients’ demand by varying lipid compositions, vesicle sizes, surface charges, etc.
-
Liposome encapsulation: we employ customized protocols to encapsulate drug molecules into liposome with high encapsulation efficiency.
-
Liposome-drug complex analysis and characterization: we can offer comprehensive analysis assays for liposomes before and after encapsulation, which includes visual appearance, size distribution, stability, Zeta potential, lamellarity, entrapment efficiency, and release rate.
Quotations and Ordering

References:
1. Rajashekar Kammari, et al. Nanoparticulate Systems for Therapeutic and Diagnostic Applications. Micro and Nano Technologies. 2017, 105-144.
2. S.Narasimha MurthyH.N.Shivakumar. Handbook of Non-Invasive Drug Delivery Systems. Personal Care & Cosmetic Technology. 2010, 1-36.
3. Neda Naseri, et al. Solid Lipid Nanoparticles and Nanostructured Lipid Carriers: Structure, Preparation and Application. Adv Pharm Bull. 2015, 5(3): 305–313.


 Figure 1. Illustration of solid lipid nanoparticles and drug loading.
Figure 1. Illustration of solid lipid nanoparticles and drug loading.