Subcutaneous Delivery
 CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. We also have developed mature technology platforms for drug delivery, such as inorganic nanomaterials, biomacromolecules, polymeric and lipid system. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy. We are delicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial researchers and assay developers all around the world. One of our focus areas is subcutaneous (SC) delivery which is defined as the administration of pharmaceutically active agents that is administered as a bolus into the subcutis to obtain local or systemic effects.
CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. We also have developed mature technology platforms for drug delivery, such as inorganic nanomaterials, biomacromolecules, polymeric and lipid system. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy. We are delicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial researchers and assay developers all around the world. One of our focus areas is subcutaneous (SC) delivery which is defined as the administration of pharmaceutically active agents that is administered as a bolus into the subcutis to obtain local or systemic effects.
Introduction to Subcutaneous Delivery
SC administration is done by injecting drugs into the fatty tissues just beneath the skin, which is the standard route of administration in diseases such as diabetes and rheumatoid arthritis. This route possesses several advantages. It is suitable for self-injection and thus improves patient compliance and reduces the cost. In addition, SC injection only takes no more than a few minutes, which is much shorter than the one needed for an intravenous (IV) infusion. That's why patients prefer SC delivery more than IV administration. Subcutaneous delivery is ideal for drugs with low oral bioavailability (e.g., insulin). Moreover, the absorption rate can be controlled by using different formulations of the drug. For example, pharmaceutical manufacturers use fast-acting and slow-acting insulin preparations to control the action time of insulin. However, this route is not appropriate for drugs with irritation to tissue because of the possibility for causing necrosis and sloughing of the skin.
There are many different solutions and suspension formulations delivered by subcutaneous route. These drugs must take effect in a small concentration due to the very small injection volume. When doing an SC administration, a small needle would be inserted into the fatty subcutaneous tissue and then the drugs would reach the bloodstream through the capillaries or the lymphatic system. To be specific, large molecule drugs usually reach the bloodstream via the lymphatic vessels. However, there is an absorption process in SC route of administration, which is slower than the IV route.
Compared to IV and intramuscular (IM) injection, SC injections offer some immediate advantages. 1) For patients requiring multiple doses, SC injection is with less painful and a wider range of alternative injection sites. 2) Due to the slower drug absorption, SC route may avoid the risks of bolus administration, including infusion reactions. 3) SC injection is much safer. For example, when injecting an infectious agent, SC route keeps infection be limited to a local. 4) SC delivery costs less than the delivery of the same drug by the IV route.
Although there are many advantages of SC route, the disadvantages and limitations of SC drug delivery still exist. Despite large development efforts, however, there are few SC-injectable protein agents on the market. This is because of the limited injection volume of SC route. When formulating products like proteins (such as monoclonal antibodies), it's hard to keep the injection volume low while the concentration is high. Besides, patients will find it difficult to inject protein products through the narrow-gauge needle due to the viscosity, solubility, and protein aggregation. And for those products with limited solubility, large volumes must be divided and injected via more frequent, small-volume injections. Moreover, at the injection site, interstitial SC fluid would trap and make locally degrade of some drugs, leading to lowered bioavailability. This, in turn, requires an increased dose (and thus volume), exacerbating the issue of limited injection volume and resulting in some of the drugs to be wasted. This reduced bioavailability also means more drugs left at the injection site, causing safety issues, including immunogenicity: irritation, vasoconstriction, abscess formation, necrosis, etc. When designing new drugs, these limitations must be carefully considered in assessing the feasibility of SC formulation development.
Recently, researchers have developed several drug delivery strategies using biocompatible nanocarriers (such as nanoparticles, liposomes, dendrimers and other micellar) to formulate human insulin, which could overcome the issue of insulin premature degradation. Researchers typically focus on developing nanoparticles for the delivery of insulin via using several natural and synthetic polymers to manufacture polymeric nanoparticles. These polymeric materials could be used to alter physicochemical characteristics of the therapeutic agents, drug release properties and biological action of the nanoparticle. Besides, using polymeric materials helps increase macromolecule stability and enhance the solubility of hydrophobic drugs.
Poly (lactic-co-glycolic acid) (PLGA) is biocompatible, biodegradable, commercially available. It has been widely used in nanotechnology applications as well as pharmaceutical formulations approved by the FDA. Moreover, poly (ethylene glycol) (PEG) could enhance drug encapsulation and modulate drug release profile to get a better sustained effect (Figure 1), and together with PLGA they can function as co-block polymer during nanoparticle preparation.
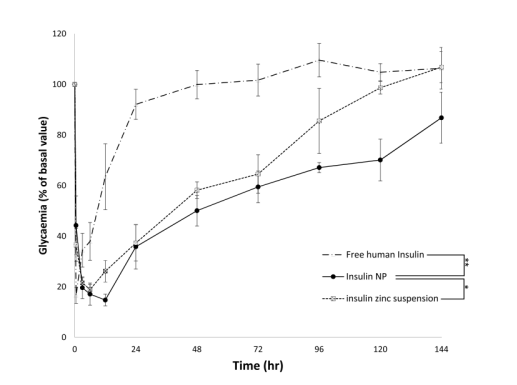
Figure 1. Profiles of glycaemia after a subcutaneous administration of free human insulin (triangles), insulin-loaded nanoparticles (circles) and insulin zinc suspension (squares) in diabetic rats. Insulin was administered at the concentration of 25 IU/kg. Before the injections, average blood glucose level was 520.28 ± 58.60 mg/dL. Results are expressed as means ± SD. (n = 6 per group). Insulin NP was statistically different from free human insulin (**p < 0.01) and insulin zinc suspension (*p < 0.05). (Abdelkader, D. H., El-Gizawy, S. A., Faheem, A. M., McCarron, P. A., & Osman, M. A. (2018). Effect of process variables on formulation, in-vitro characterization and subcutaneous delivery of insulin PLGA nanoparticles: An optimization study. Journal of Drug Delivery Science and Technology, 43, 160–171)
Our Featured Services
CD Bioparticles is specialized in the development of drug delivery systems and customizing nanoparticles for drug delivery utilizing our core technologies. With our high-quality products and services, the efficacy of your drug delivery can be tremendously improved.
We offer custom synthesis of polymer microspheres and nanoparticles. Clients may select the material type, particle size, size distribution, color dye, fluorescent dye, and/or surface functional groups such as carboxyl or amine groups. We also encapsulate proteins, steroids, ligands, nucleic acids, drug molecules, and other agents. Additionally, coating the surface of your microspheres or nanoparticles with ligands, oligonucleotides, and other agents are available to us.
References:
1. Dychter, S. S., Gold, D. A., & Haller, M. F. (2012). Subcutaneous drug delivery. Journal of Infusion Nursing, 35(3), 154–160.
2. Abdelkader, D. H., El-Gizawy, S. A., Faheem, A. M., McCarron, P. A., & Osman, M. A. (2018). Effect of process variables on formulation, in-vitro characterization and subcutaneous delivery of insulin PLGA nanoparticles: An optimization study. Journal of Drug Delivery Science and Technology, 43, 160–171.
3. Richter, W. F., & Jacobsen, B. (2014). Subcutaneous absorption of biotherapeutics: knowns and unknowns. Drug Metabolism and Disposition, 42(11), 1881–1889.


 CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. We also have developed mature technology platforms for drug delivery, such as inorganic nanomaterials, biomacromolecules, polymeric and lipid system. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy. We are delicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial researchers and assay developers all around the world. One of our focus areas is subcutaneous (SC) delivery which is defined as the administration of pharmaceutically active agents that is administered as a bolus into the subcutis to obtain local or systemic effects.
CD Bioparticles is a leading manufacturer and supplier of various drug delivery products, including metal nanostructures, biomacromolecules, synthetic polymer and biopolymers and lipid system, for R&D and commercialization in a variety of application areas. We also have developed mature technology platforms for drug delivery, such as inorganic nanomaterials, biomacromolecules, polymeric and lipid system. In addition, we can offer a wide range of custom services including drug delivery nanoparticles formulation, bioparticles analysis and characterization, and drug targeting strategy. We are delicated to providing the most comprehensive list of products and fit-for-purpose custom analysis and synthesis services to academia as well as industrial researchers and assay developers all around the world. One of our focus areas is subcutaneous (SC) delivery which is defined as the administration of pharmaceutically active agents that is administered as a bolus into the subcutis to obtain local or systemic effects.
