Sustained-Release Microspheres
Introduction of Sustained-Release Microspheres
Sustained-release microspheres have gained significant interest in the pharmaceutical industry in recent years due to their potential to improve drug efficacy and patient compliance. CD Bioparticles is a leading formulation specialist company that has over 10 years of experience in the industry.
Our Membrane Emulsion Sedimentation (MES)Technology
The membrane emulsion sedimentation microsphere preparation process developed by our research and development team has the advantages of both membrane emulsification and microfluidic methods, avoiding their respective shortcomings while achieving continuous microsphere production. Microsphere production cannot be enlarged based on the current regulatory standards, which greatly increases the development cost of microsphere formulations. Our membrane emulsion sedimentation microsphere production process fills this gap in microsphere production technology. Microspheres can be produced from pilot scale to production scale without changing the device, relying on the extension of production time.
This technology offers numerous advantages, such as the ability to produce microspheres with a narrow size distribution, excellent batch-to-batch reproducibility, and a high drug loading capacity.
At CD Bioparticles, we have extensive experience in membrane emulsion sedimentation technology, enabling us to produce high-quality sustained-release microspheres for our clients.
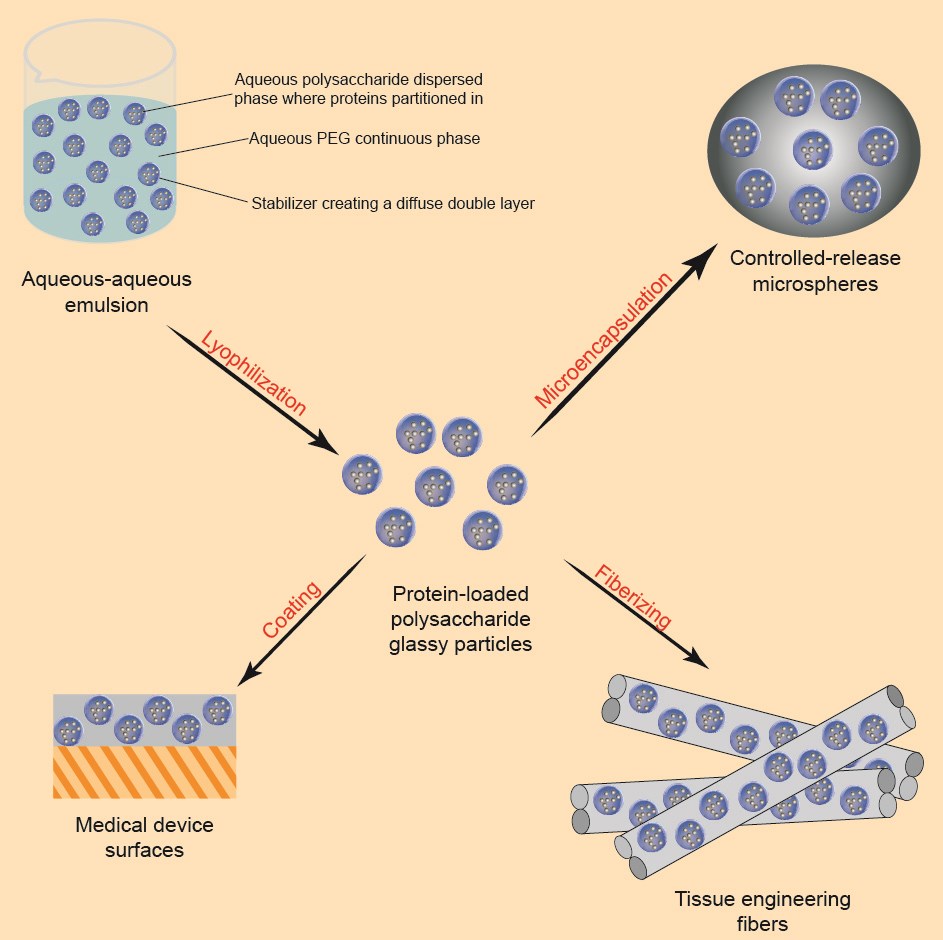
Our Water-in-Water Emulsion Technology
Currently, there are no protein drug microsphere formulations on the market, and there have been microsphere formulations of somatotropin that were withdrawn from the market due to technical problems. Our scientists invented the "Water-in-Water Emulsion" protein premix technology, which can encapsulate proteins in polysaccharide particles without contact with organic solvents, fix their folding method, and then use Our continuous microsphere production equipment to prepare microspheres. This technology has been confirmed not to undergo conformational changes and does not produce harmful antibodies in monkeys.
Manufacturing Capability and Quality
CD Bioparticles has state-of-the-art facilities for the production of sustained-release microspheres using MES technology. Our manufacturing process is GMP-like compliant, and we have strict quality control procedures to ensure the highest product quality.
We have the capability to produce sustained-release microspheres in a range of sizes, from nanometers to micrometers, and with different drug release profiles, such as zero-order and first-order release kinetics. Our team of experienced scientists and engineers can work with clients to develop custom formulations that meet their specific needs.
-
It can achieve a production capacity of more than 1,000 microspheres per batch;
-
Aseptic production conditions;
-
It meets the basic standards for clinical phase I trial drugs.
Suitable APIs
Currently, nearly 200 protein drugs and nearly 100 peptide drugs have been approved for marketing. Our sustained-release microsphere technology is suitable for dozens or even hundreds of drugs, not limited to individual or a few products. Our research and development pipeline includes several protein microspheres and dozens of peptide drugs at different stages of basic research to industrial development. The role of these products is not only to develop products, but also to demonstrate Our platform technology and form collaboration in microsphere development.
CD Bioparticles has experience working with a wide range of APIs, including peptides, proteins, small molecules, and nucleic acids. We can provide consultation services to help clients select the most suitable API for their formulation.
Our Microspheres Advantages
CD Bioparticles is a leading provider of sustained-release microspheres, with a proven track record of success in the industry. Our membrane emulsion sedimentation technology and water in water technology allow us to produce high-quality microspheres with a range of APIs, and our experienced team of professionals is dedicated to providing our clients with the best possible service. Whether you are looking for assistance with formulation development, scale-up and manufacturing, or quality control, CD Bioparticles is the ideal.


