|
|
Category
|
Typical Carrier-surface Material / Decoration
|
Main mechanism
|
|
Mucus adhesive
|
Synthetic polymer
|
Poly (lactic acid) (PLA), Poly (sebacic acid) (PSA), Poly (lacticacid-glycolic acid) (PLGA), Poly (acrylic acid) (PAA), Carbopol, Polycarbophil, Copolymer of methylvinylether and methacrylic acid, and their Derivatives
|
Hydrogen bonding, Electrostatic interaction, Long-chain entanglement
|
|
Cellulose derivative
|
Carboxy methyl cellulose (CMC), Thiolated carboxy methyl cellulose (TCMC), Sodium carboxy methyl cellulose (SCMC), Hydroxyethyl cellulose (HEC), Hydroxypropyl cellulose (HPC), Methyl hydroxyethyl cellulose (MHPC), Methyl cellulose (MC), Hydroxypropyl methyl- cellulose (HPMC)
|
Hydrogen bonding, Electrostatic interaction, Long-chain entanglement
|
|
Natural polysaccharide
|
Chitosan, Gelatin, Hyaluronic acid, Carrageenan, Pectin, Sodium alginate and their derivatives
|
Hydrogen bonding, Electrostatic interaction, Long-chain entanglement
|
|
Natural protein
|
Keratose, Kerateine
|
Hydrogen bonding, Electrostatic interaction, Covalent disulfide bond
|
|
Sulfhydrylation agent
|
Cysteine, Thioglycolic Acid (TGA), 4-Thiobuthylamidine (TBA), N-acetyl-cysteine, Isopropyl-S-acetyl thioacetimidate, Glutathione
|
Covalent disulfide bond
|
|
Mucus penetration
|
Synthetic polymer
|
Polyethylene glycol (PEG), Pluronic F127 (PF127), Poly(2-hydroxypropyl) methacrylamide (PHPMA), Polysarcosine (PSAR), Poly(vinyl alcohol) (PVA), Poly(2-alkyl-2-oxazoline) (PAOXA), Hydroxyl-containing non-ionic water-soluble polymers, Zwitterionic polymers (polybetaines)
|
Shielding electrostatic and hydrophobic interactions
|
|
Small molecular compound
|
N-acetylcysteine, 2-mercapto-N-octylacetamide, N-dodecyl-4-mercaptobutylamide
|
Destroying mucin disulfide bond
|
|
Proteolytic enzyme
|
Papain, Bromelain, Trypsin
|
Hydrolyzed mucin
|
|
Promote transcellular transport
|
Penetration
enhancer
|
Nonionic surfactants (such as Pluronic P123, F68 and F127)
|
Alter cell membrane fluidity Decrease transepithelial
resistance; Inhibit the effect of P-glycoprotein efflux
|
|
Bile salts and derivatives
|
Alter cell membrane fluidity; Interact with bile acid transporter to induce endocytosis and uptake
|
|
Medium-chain fatty acids and derivatives
|
Alter cell membrane fluidity; Decrease transepithelial resistance
|
Positively
charged polymer
|
Chitosan and derivatives
|
Reversible decrease of the transepithelial resistance; Electrostatic interaction with cell surface prolongs absorption time
|
Cell penetrating
peptide
|
HIV-1 Tat, penetratin, oligoarginine, MAP and R8
|
Assist carriers enter directly into cells through lipid membrane or be internalized by endocytosis.
|
|
Broad enterocyte surface targeting ligand
|
IgG (for Neonatal Fc receptor (FcRn, FCGRT)); Lactoferrin (for Lactoferrin receptor (ITLN-1)); Vitamin B12 (for Vitamin B12–intrinsic factor receptor (CUBN, AMN))
|
Trigger transcytosis mechanism of receptors in cell membrane
|
|
M cell targeting ligand
|
Lectin (UEA-1, WGA, TL and LTA)
|
Combine with cell surface glycoproteins and sugar esters to prolong absorption and promote endocytosis
|
|
HA recombinant proteins whose C terminus are introduced with CPE30 (the terminal 30 amino acids of Clostridium Perfringens enterotoxin)
|
Bound to claudin-4 receptor that is highly expressed in M cells
|
|
RGD and LDV peptidomimetics
|
Bound to integrins on the apical surface
of M cells
|
|
Promote paracellular transport
|
Penetration
enhancer
|
Nonionic surfactants, bile salts, medium-chain fatty acids and their derivatives
|
Irreversibly dissolve cell membrane components Combine with extracellular Ca2+ to induce redistribution of target proteins of calmodulin
|
Positively
charged polymer
|
Chitosan and derivatives
|
Possible mechanism is reversible interaction with tight junction proteins
|
|
Peptide
|
ADT-6 HAV-6 C-CPE 7-mer(FDFWITP or PN-78) AT-1002 PN159(KLAL or MAP)
|
Interacting with occludin and claudins to regulate the tight junction opening
|
|
Myosin light chain phosphatase (MLCP) inhibitory peptides 640 and 250 (synthetic peptides emulating interfacial contacts involving MLCP regulatory proteins CPI-17 and MYPT)
|
Inhibit dephosphorylation catalyzed by MLCP to prevent reversion of opened tight junction
|
|
Bacterial protein
|
Clostridium perfringens enterotoxin (CPE)
|
Binding to claudins to regulate the tight junction opening
|
|
Metal chelating agent
|
Citrate
|
Combine with extracellular Ca to induce redistribution of target proteins of calmodulin
|


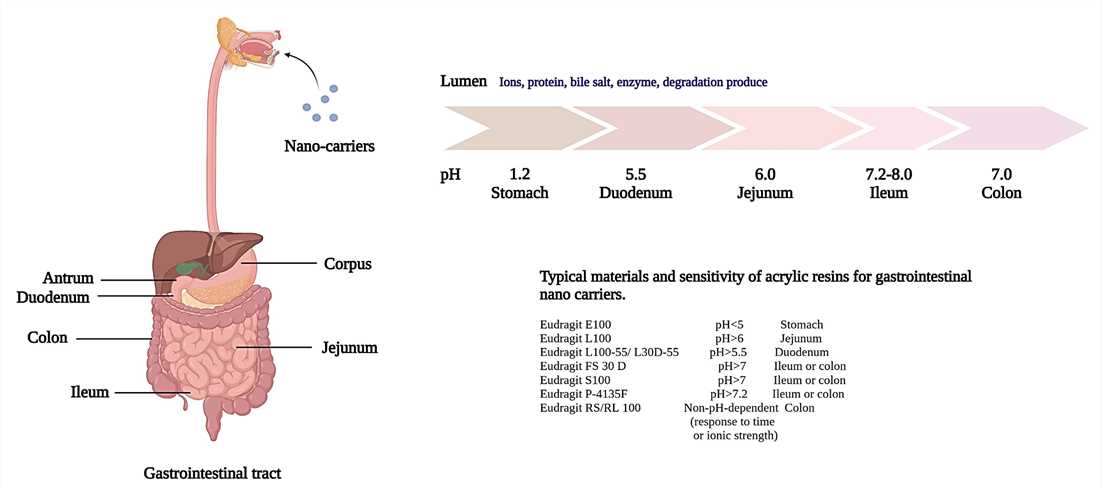 Figure 1. Schematic model of gastrointestinal tract. Each segment has different pH values and epithelial characteristics.
Figure 1. Schematic model of gastrointestinal tract. Each segment has different pH values and epithelial characteristics.
