Toxicity Measurement
CD Bioparticles now provide various strategies for drug toxicity study. Our experienced scientists are dedicated to work on efficient and economically feasible solutions in order to establish a comprehensive platform to measure the toxicity of drugs loaded in different bioparticles.
Toxicity Measurement Introduction
Nanoparticles are used as carriers for drug delivery, and their own safety to organisms or cells is receiving increasing attention. As a drug carrier, in addition to the nature of the carrier, it also has less toxicity to the organism. In this way, the side effects of the drug carried by nanoparticle can be effectively reduced, and the applicability of the drug delivery system can be improved. Therefore, the assessment of the toxicity of nanoparticles for transporting drugs is an essential parameter for the construction of nanoparticle drug delivery systems. Various methods are available for the toxicity assessment, which can be categorized as In vitro and in vivo. Generally, In vitro methods used to assess nanoparticle toxicity fall into two general categories: functional assays and viability assays. Functional assays are those that seek to assess the effects of nanoparticles on various cellular processes, whereas viability assays are concerned solely with whether a given nanoparticle causes death in a cell or a system of cells. The in vivo toxicity assessment is normally performed on animal models such as mice and rats.
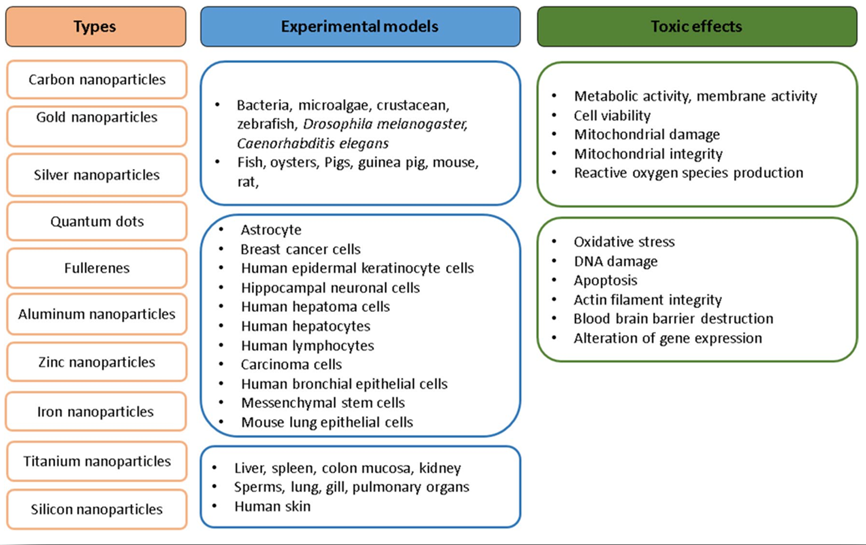
Figure 1. Nanoparticles types, experimental models used for the studies and toxic effects of nanoparticles. (Vinay K., et al. International Nano Letters. 2017, 7:243–256)
Toxicity Measurement Analysis Methods
In Vitro Assessment Assays
The functional assays that seek to assess the effects of nanoparticles on various cellular processes DNA synthesis are introduced below from 1-7. Viability assays include 8-10.
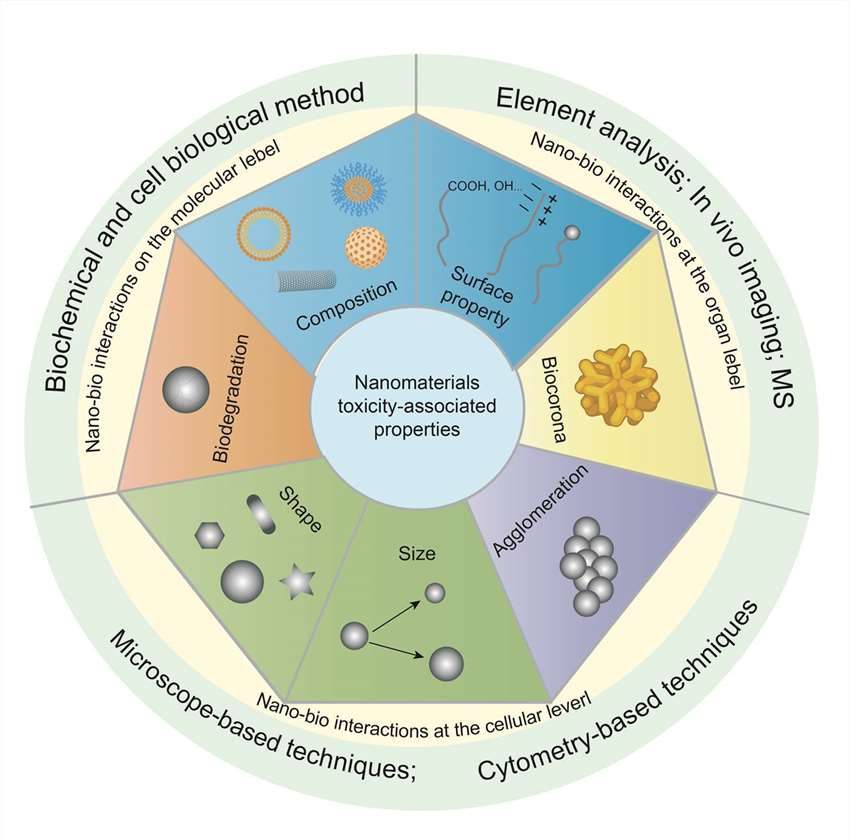 Figure 2. Factors affecting nanoparticle toxicity.
Figure 2. Factors affecting nanoparticle toxicity.
1. DNA Synthesis
DNA synthesis assays give critical information about the proliferative state and general health of dividing cells. The incorporation of BrdU (5-bromo- 2-deoxyuridine) into newly synthesized DNA has been frequently employed to quantify DNA synthesis in nanotoxicity assays.
2. DNA Damage
Damage to DNA is a fundamental example of cellular toxicity, and it is critical to assess such damage caused by any nanoparticle, given that damage to DNA is highly correlated with an increased risk of cancer. The measurement of number of single-strand breaks in DNA is the most common method to assess DNA damage by single-cell gel electrophoresis assay.
3. Altered Gene Expression
Understanding the effect of nanoparticles on the cellular genome is a critical step toward achieving a real understanding of any nanoparticle’s toxicity profile. The activity of functional genes implicated in various cellular processes can be quantitatively assessed by DNA microarray (general) or polymerase chain reaction (PCR; specific) analysis.
4. Immunogenicity
The ability of a given nanoparticle to evoke an immune response is a critical indicator of its toxicity to physiological systems. Cytokine levels can be accurately detected at minute volumes (picograms per milliliter) by an enzyme-linked immunosorbent assay.
5. Oxidative Stress
An increase in the presence of ROS in the cellular environment has the potential to damage or disrupt key cellular processes of a host. The increase in ROS may result either from an innate immune response to a nanoparticle or from the ability of a specific nanoparticle (e.g., a fullerene or a metal oxide) to autocatalyze ROS formation. Generally, the presence of ROS is assessed either directly (by quantifying the amount of ROS present in a given cell population) or indirectly (by monitoring the secondary effects of prolonged oxidative stress).
6. Cell Proliferation
The rate of cell growth is an important indicator of overall cell health and of the potential for nanoparticles to interfere with proliferative processes. Two quantitative assays have emerged as the standard for assessing cell proliferation: (a) cell counting by flow cytometry or high-content image analyzers and (b) the colony-forming efficiency (CFE) assay.
7. Exocytosis
The effect of nanomaterials on vital cellular processes, such as exocytosis, should be intimately understood before any nanomaterial is deemed safe. Carbon-fiber microelectrode amperometry has been employed to study the effect of various nanoparticles on the secretion of electroactive small molecules (e.g., serotonin, epinephrine).
8. Metabolic Activity
By a considerable margin, assays of metabolic activity are the most common methods used to determine cell viability following nanoparticle exposure. Of these assays, the most popular is the MTT assay. In live cells, MTT [3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide] is reduced to purple formazan, which can be detected spectrophotometrically.
9. Hemolysis
The lysis of erythrocytes in response to nanoparticles can be a measure of both membrane disruption and extreme cellular toxicity (i.e., necrosis) and is especially important for nanoparticles that are intended to be directly introduced into the bloodstream. The spectrophotometric detection of hemoglobin is an extremely sensitive technique for the detection of hemolysis.
10. Apoptosis and Necrosis
Measurements of the indicators of programmed cell death (i.e., apoptosis) and/or necrosis directly reveal nanoparticles’ ability to induce intracellular suicide mechanisms or destroy cells. Such assays focus largely on measuring membrane integrity, but some also attempt to measure apoptotic protein levels/activation and DNA fragmentation. The main techniques are used to determine membrane integrity: phosphatidylserine (which migrates to the extracellular surface of apoptotic cells) labeling with annexin V, propidium iodide exclusion by intact membranes.
In Vivo Assessment Assays
The in vivo tests are essentially involved in implanting the nanoparticles into an animal model and the evaluation of its histocompatibility. Dose and LD50, effects on organ systems could be measured accordingly for toxicity assessment.
CD Bioparticles offers a full set of service for bioparticle toxicity measurement. For more detailed information, please feel free to contact us or directly send us an inquiry.
Quotations and Ordering

References:
1. Sara A. L., et al. Assessing Nanoparticle Toxicity. Annu. Rev. Anal. Chem. 2012, 5:181–205.
2. Haji B., et al. Toxicity of Nanoparticles and an Overview of Current Experimental Models. Iranian Biomedical Journal. 2016, 20(1): 1-11.
3. Vinay K., et al. In vitro and in vivo toxicity assessment of nanoparticles. International Nano Letters. 2017, 7:243–256.
4. Sara A. L., et al. Examples of functional used to assess nanoparticle toxicity. Annu. Rev. Anal. Chem. 2012, 5:181–205.



 Figure 2. Factors affecting nanoparticle toxicity.
Figure 2. Factors affecting nanoparticle toxicity.