Vaginal Administration
Introduction of Vaginal Administration
Medication can be administered vaginally, passing through the vagina. It may be found in creams, gels, tablets, suppositories, and rings, among other forms. Utilizing vaginal to give medication has a number of benefits. The fact that the drug is delivered directly to the affected area is one of its most important advantages. With this localized and focused strategy, the medicine may be more effective at higher doses. Another benefit is that it does not require needles or injections because it is non-invasive. Patients can also give their own medication, which is more convenient and comfortable for them.
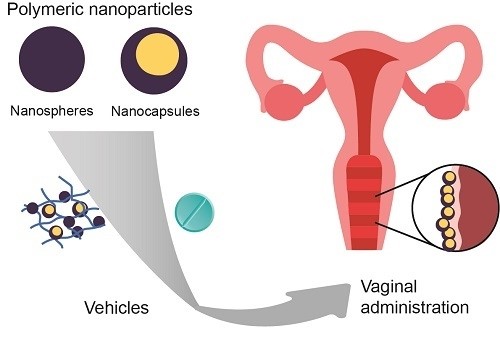 Figure 1. Vaginal Administration. (Leyva-Gómez G, et al.; 2018)
Figure 1. Vaginal Administration. (Leyva-Gómez G, et al.; 2018)
However, there are also a few drawbacks to vaginal administration. Its potential to occasionally produce irritability or discomfort is one of its most prevalent downsides. This is due to the vaginal lining's sensitivity and potential for adverse drug reactions. Additionally, if the medication is not sterile or if the area already has infections, vaginal administration can increase the risk of infections.
The Application of Vaginal Administration
-
Diseases for which Vaginal Administration Applies
Many illnesses, especially those that impact the reproductive system, are acceptable for vaginal administration. Yeast infections, bacterial vaginosis, and sexually transmitted infections are among the illnesses for which it is frequently used. Hormone drugs are also administered vaginally to treat menopause, other reproductive health concerns, and contraception. Additionally, the vaginal delivery of medications for other illnesses like HIV and cancer is currently being studied.
-
Vaginal Administration's Nano-delivery Product FDA or Clinical Case
Nano-delivery solutions for vaginal administration have multiple FDA-approved clinical instances. One such is the antifungal drug VT-1161, created by Mycovia Pharmaceuticals. Nanoparticles that surround VT-1161 boost its bioavailability and enable the administration of a lower dose. A vaginal ring that releases the hormone etonogestrel for contraception is another illustration. Over the course of three weeks, the hormone is gradually released from the ring, offering minimal adverse effects and effective contraception.
Characteristics of Vaginal Administration
-
Biobarriers for Vaginal Administration
The cervix and the vaginal epithelium serve as the biological barriers to vaginal administration. These barriers may restrict the amount of medication that enters the bloodstream, which may lessen the systemic effects of the medication. The vaginal epithelium is made up of numerous layers of cells, each of which has a different level of permeability. It is more difficult for medication to penetrate the epithelium close to the cervix than the epithelium close to the vaginal entrance.
-
The Advantages of Nanoparticles in Vaginal Administration
Nanoparticles are beneficial for vaginal administration in a number of ways. They can improve the solubility and bioavailability of medications, which is one of their key advantages. By using a lesser dose of the medicine, the chance of side effects can be decreased. Additionally, nanoparticles can increase the stability of a drug and shield it against deterioration, extending its shelf life. Nanoparticles can also assist direct medication to particular tissues, such the vaginal epithelium, which can boost effectiveness and lessen unwanted effects.
Our Featured Services
CD Bioparticles is specialized in the development of drug delivery systems and customizing nanoparticles for drug delivery utilizing our core technologies. With our high-quality products and services, the efficacy of your drug delivery can be tremendously improved.
We offer custom synthesis of polymer microspheres and nanoparticles. Clients may select the material type, particle size, size distribution, color dye, fluorescent dye, and/or surface functional groups such as carboxyl or amine groups. We also encapsulate proteins, steroids, ligands, nucleic acids, and other drug molecules. Additionally, the surface coating of microspheres or nanoparticles with ligands, oligonucleotides, and other agents are available for clients to choose.
References
-
Buzzaccarini G, et al.; Hyaluronic acid in vulvar and vaginal administration: evidence from a literature systematic review. Climacteric. 2021, 24(6):560-571.
-
Alexander NJ, et al.; Why consider vaginal drug administration? Fertil Steril. 2004, 82(1):1-12.
-
da Silva PB, et al.; Nanotechnological strategies for vaginal administration of drugs--a review. J Biomed Nanotechnol. 2014,10(9):2218-43.
-
El-Hammadi MM, Arias JL. Nano-sized platforms for vaginal drug delivery. Curr Pharm Des. 2015, 21(12):1633-44.
-
Leyva-Gómez G, et al.; Approaches in Polymeric Nanoparticles for Vaginal Drug Delivery: A Review of the State of the Art. Int J Mol Sci. 2018, 19(6):1549.


 Figure 1. Vaginal Administration. (Leyva-Gómez G, et al.; 2018)
Figure 1. Vaginal Administration. (Leyva-Gómez G, et al.; 2018)