Liposomes vs. Biodegradable Polymers in Drug Delivery: Which Platform Delivers More Value
Drug delivery is no longer just about transporting an active pharmaceutical ingredient (API) from point A to point B. Modern formulation scientists are tasked with improving bioavailability, extending circulation time, minimizing toxicity, and ensuring reproducible manufacturing. Among the most widely adopted nanocarrier systems, liposomes and biodegradable polymer-based nanoparticles remain at the center of formulation strategy discussions.
While both platforms have proven clinical and commercial success, their performance can vary significantly depending on the drug molecule, therapeutic goal, and route of administration. Recent comparative studies across nutrients, antioxidants, and anti-inflammatory compounds provide valuable insights into how these systems behave in real-world applications. This article examines liposomes and biodegradable polymers side by side, using published evidence to clarify their strengths, limitations, and ideal use cases.
Understanding the Two Delivery Platforms
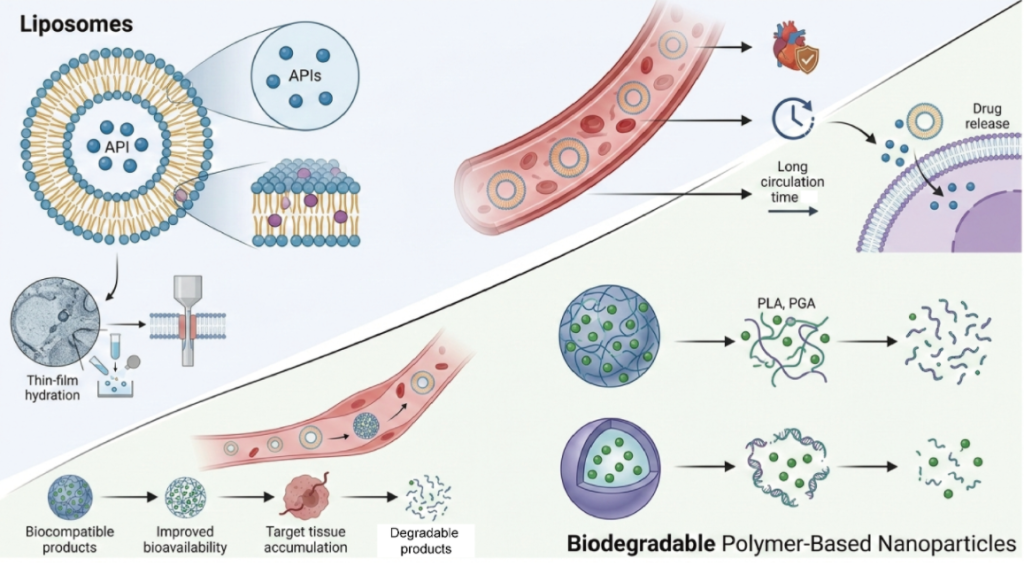
Liposomes: Biomimicry with Clinical Legacy
Liposomes are spherical vesicles composed of phospholipid bilayers that closely resemble biological membranes. This structural similarity allows them to encapsulate both hydrophilic and lipophilic drugs while maintaining excellent biocompatibility.
They have a long regulatory history, with multiple FDA-approved formulations in oncology, vaccines, and infectious disease treatment. Liposomes are often selected when cell membrane compatibility, safety, and rapid clinical translation are key considerations.
Biodegradable Polymers: Engineered Precision and Control
Biodegradable polymer nanoparticles—commonly based on PLGA, PLA, or chitosan—offer greater structural rigidity and tunable degradation kinetics. Drug release can be precisely controlled by adjusting polymer composition, molecular weight, and particle architecture.
These systems are increasingly favored when long-term release, improved intracellular uptake, or enhanced stability is required, particularly for small molecules and nutraceutical compounds.
Particle Size, Uniformity, and Stability: What the Data Shows
A comparative study published in AAPS PharmSciTech (2022) evaluated liposomal L-carnitine (lipo-carnitine) against PLGA-based nano-carnitine formulations.
The results revealed clear physicochemical differences:
- Particle size: Liposomes formed smaller particles (~98 nm), while polymeric nanoparticles were larger (~251 nm).
- Dispersion quality: Despite their larger size, polymer nanoparticles exhibited a lower aggregation index, indicating more uniform particle distribution and improved colloidal stability.
This distinction is particularly relevant for formulations requiring long shelf life or consistent batch-to-batch performance, where aggregation can compromise efficacy.
Release Kinetics and Half-Life Extension
Rapid clearance remains a common challenge for free drug molecules. In the same L-carnitine study, free L-carnitine released nearly 90% of its payload within one hour, whereas both liposomal and polymer-based carriers sustained drug release for up to 12 hours.
However, polymer nanoparticles demonstrated slightly tighter control over release profiles, which can translate into more predictable pharmacokinetics, especially for chronic or metabolic indications.
This sustained release behavior highlights why biodegradable polymers are often chosen for formulations where dosing frequency reduction is a key objective.
Encapsulation and Drug Loading Efficiency
Encapsulation efficiency directly affects manufacturing cost and therapeutic consistency. In the L-carnitine comparison, polymeric nanoparticles showed a modest advantage over liposomes in both drug loading and encapsulation efficiency.
This advantage becomes more pronounced for small, water-soluble molecules, where liposomes may suffer from leakage during storage or circulation. Polymer matrices, by contrast, physically entrap the drug, reducing premature loss.
Bioavailability in Human Applications: Vitamin B12 as a Case Study
A 2023 clinical study published in the Journal of Pharmacology and Toxicology compared vitamin B12 delivered via nanoparticles, liposomes, and ester-based formulations.
The findings were unambiguous:
- Nanoparticle delivery achieved the highest serum B12 levels
- Liposomal delivery showed lower absorption efficiency
- Ester formulations demonstrated intermediate performance
Moreover, nanoparticle-based B12 exhibited reduced rapid clearance, maintaining therapeutic blood concentrations for longer periods.
This study underscores a critical consideration for oral and systemic delivery: structural stability and metabolic protection often outweigh membrane mimicry when absorption and retention are priorities.
Cellular Uptake and Functional Performance: Antioxidant Delivery
Not all delivery challenges are systemic. Intracellular uptake is crucial for antioxidants and cytoprotective agents. An in vitro study evaluated epicatechin-loaded liposomes and PLA nanoparticles.
Key observations included:
- Higher cellular uptake with polymer nanoparticles (36.29%) compared to liposomes (27.05%)
- Both systems protected renal tubular cells from oxidative damage
- Polymer nanoparticles achieved stronger protection at lower concentrations
These results suggest that polymer carriers may offer a therapeutic efficiency advantage when intracellular delivery is required, reducing the total dose needed to achieve biological effects.
Anti-Inflammatory Drug Delivery: Biocompatibility vs. Stability
A 2024 study in the International Journal of Molecular Sciences compared liposomes with chitosan-based magnetic nanocapsules for anti-inflammatory drug delivery.
Both platforms demonstrated excellent biocompatibility, reinforcing their suitability for biomedical use. However, polymer-based nanocapsules provided a more stable delivery environment under specific experimental conditions, which can be critical for sensitive anti-inflammatory compounds.
This added stability can be especially valuable in complex biological environments where pH changes or enzymatic activity may compromise lipid-based systems.
Choosing the Right System: Strategic Considerations
Rather than framing liposomes and biodegradable polymers as competitors, the evidence points to complementary strengths:
- Liposomes excel in safety, biomimicry, and rapid clinical translation
- Biodegradable polymers offer superior control over release, stability, and cellular uptake
The optimal choice depends on factors such as drug chemistry, target tissue, route of administration, and desired release profile. In many advanced formulations, hybrid or sequential strategies may even combine both platforms.
Final Thoughts
As drug delivery science continues to evolve, formulation decisions are increasingly driven by data-driven comparisons rather than tradition. Recent studies across nutrients, antioxidants, and anti-inflammatory agents consistently show that biodegradable polymer nanoparticles often outperform liposomes in stability, bioavailability, and intracellular delivery—while liposomes remain indispensable where biocompatibility and regulatory familiarity are paramount. Understanding these nuanced differences enables more informed formulation strategies, ultimately leading to safer, more effective therapeutic products.
Related Products
Liposomes
| Product Name | Catalog | Lipid composition | Liposome Size | |
| Clipos™ Cardiolipin Lipids (CL) Liposomes | CDECAR-1644 | PC/CL (1/99 molar ratio) | 100 nm | Buy Now |
| Clipos™ Cardiolipin Lipids (CL) Liposomes | CDECAR-1645 | PC/CL (0.5/99.5 molar ratio) | 100 nm | Buy Now |
| Clipos™ Cardiolipin Lipids (CL) Liposomes | CDECAR-1648 | 100% 14/0 Cardiolipin (TMCL) | 50 nm | Buy Now |
| Clipos™ Cardiolipin Lipids (CL) Liposomes | CDECAR-1649 | PC/CL (90/10 molar ratio) | 50 nm | Buy Now |
| Clipos™ DOTAP Liposomes | CDECDEP-1647 | DOTAP/DOPC (40/60 molar ratio) | 100 nm | Buy Now |
| Clipos™ DOTAP Liposomes | CDECDEP-1648 | DOTAP/DOPC (30/70 molar ratio) | 100 nm | Buy Now |
| Clipos™ DOTAP Liposomes | CDECDEP-1649 | DOTAP/DOPC (20/80 molar ratio) | 100 nm | Buy Now |
| Clipos™ DOTAP Liposomes | CDECDEP-1650 | DOTAP/DOPC (10/90 molar ratio) | 100 nm | Buy Now |
| Clipos™ DOTAP Liposomes | CDECDEP-1651 | DOTAP/DOPC (5/95 molar ratio) | 100 nm | Buy Now |
| Clipos™ Diether Phosphatidylcholine (PC) Liposomes | CDECDEE-1610 | 100% 12/0 Diether PC | 100 nm | Buy Now |
| Clipos™ Diether Phosphatidylcholine (PC) Liposomes | CDECDEE-1611 | 100% 13/0 Diether PC | 100 nm | Buy Now |
| Clipos™ Diether Phosphatidylcholine (PC) Liposomes | CDECDEE-1612 | 100% 14/0 Diether PC | 100 nm | Buy Now |
| Clipos™ Diether Phosphatidylcholine (PC) Liposomes | CDECDEE-1613 | 100% 16/0 Diether PC | 100 nm | Buy Now |
| Clipos™ Diether Phosphatidylcholine (PC) Liposomes | CDECDEE-1614 | 100% 18/0 Diether PC | 100 nm | Buy Now |
| Clipos™ Phosphatidylglycerol (PG) Liposomes | CDECPG-1656 | DOPG/DOPC (90/10 molar ratio) | 100 nm | Buy Now |
| Clipos™ Phosphatidylglycerol (PG) Liposomes | CDECPG-1657 | DOPG/DOPC (80/20 molar ratio) | 100 nm | Buy Now |
| Clipos™ Phosphatidylglycerol (PG) Liposomes | CDECPG-1658 | DOPG/DOPC (70/30 molar ratio) | 100 nm | Buy Now |
| Clipos™ Phosphatidylglycerol (PG) Liposomes | CDECPG-1659 | DOPG/DOPC (60/40 molar ratio) | 100 nm | Buy Now |
| Clipos™ Phosphatidylglycerol (PG) Liposomes | CDECPG-1661 | DOPG/DOPC (40/60 molar ratio) | 100 nm | Buy Now |
| Clipos™ Phosphatidylglycerol (PG) Liposomes | CDECPG-1663 | DOPG/DOPC (20/80 molar ratio) | 100 nm | Buy Now |
| Clipos™ Natural Phosphatidylserine (PS) Lipid Liposomes | CDECPS-1606 | Soy PS/Cholesterol (70/30 molar ratio) | 100 nm | Buy Now |
| Clipos™ Phosphatidylserine (PS) Liposomes | CDECPS-1670 | DOPS/DOPC (90/10 molar ratio) | 100 nm | Buy Now |
| Clipos™ Phosphatidylserine (PS) Liposomes | CDECPS-1671 | DOPS/DOPC (80/20 molar ratio) | 100 nm | Buy Now |
| Clipos™ Phosphatidylserine (PS) Liposomes | CDECPS-1672 | DOPS/DOPC (70/30 molar ratio) | 100 nm | Buy Now |
| Clipos™ Phosphatidylserine (PS) Liposomes | CDECPS-1673 | DOPS/DOPC (60/40 molar ratio) | 100 nm | Buy Now |
| Clipos™ Phosphatidylserine (PS) Liposomes | CDECPS-1675 | DOPS/DOPC (40/60 molar ratio) | 100 nm | Buy Now |
