mRNA-LNP Vaccines on the Frontline of Cancer Immunotherapy
Cancer ranks as the second deadliest disease which affects people worldwide. The current cancer treatment methods including surgery and radiotherapy and chemotherapy present multiple drawbacks because they fail to remove all cancer cells and cause substantial damage to healthy tissues and lead to frequent tumor relapses. The medical field now uses cancer vaccines as an innovative therapeutic method within tumor immunotherapy research. The antigens from cancer vaccines which contain tumor-associated antigens (TAAs) and tumor-specific antigens (TSAs) become visible to major histocompatibility complex I/II (MHC I/II) after injection which activates cytotoxic T lymphocytes (CTLs) and helper T cells (Th) cells. The antigen recognition ability of the immune system becomes stronger through this process which leads to effective tumor control and metastasis prevention.
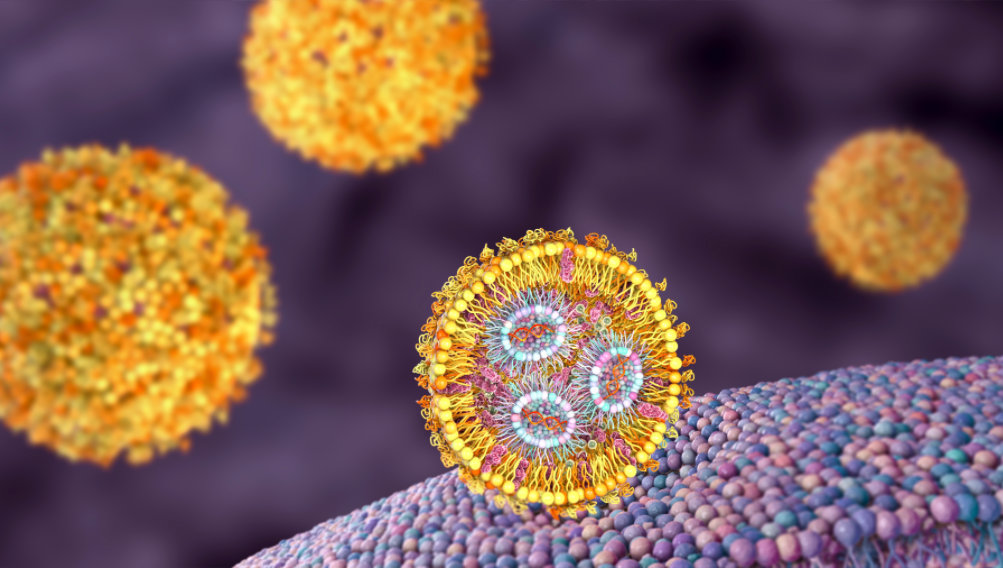
The main components of traditional vaccines consist of either inactivated viruses or recombinant proteins. The novel coronavirus pandemic has made mRNA vaccines the leading focus of modern vaccine development because they demonstrate exceptional results. The genetic information transmission properties of mRNA enable direct protein synthesis from its template sequence. The antigen production process in human cells becomes possible through mRNA vaccines which introduce specific antigen-encoding mRNA to cells for autonomous antigen production and subsequent immune system activation. The advancement of mRNA technology has made mRNA vaccines popular for disease prevention and treatment applications especially in cancer therapy. The mRNA vaccines show high individual performance but researchers combine them with other immunotherapies to boost their overall effectiveness. Multiple mRNA-based tumor immunotherapies now participate in clinical trials where they have shown promising results.
What are mRNA-LNP Vaccines?
The delivery of mRNA molecules depends on cancer vaccines for their operation. The body degrades naked mRNA molecules through ribonucleases (RNases) because these molecules lack stability. The negative charge of mRNA molecules creates a barrier to penetration through host cell membranes which reduces the efficiency of cell transfection. The delivery of mRNA through technology has experienced substantial development during the past thirty years. The delivery of mRNA through lipid nanoparticles (LNPs) represents the most advanced system currently used in clinical applications. The formulations of these systems include four essential elements which are ionizable lipids together with phospholipids and cholesterol and PEG-coated lipids. The positively charged state of ionizable lipids at low pH enables them to bind mRNA and form LNPs efficiently. The ionizable lipids maintain neutral charges when exposed to physiological conditions which leads to reduced LNP toxicity. The acidic environment of endosomal linings triggers ionizable lipid protonation which leads to LNP disruption and mRNA release. The phospholipid composition of LNPs helps maintain their stability by duplicating cell membrane structures and cholesterol controls the lipid bilayer’s fluidity. The PEGylation of lipids improves LNPs’ stability in both colloidal and serum environments which decreases their likelihood of immune system detection and removal. The success of mRNA delivery depends heavily on ionizable lipids because they determine both mRNA encapsulation rates and endosomal release which affects the final protein production levels. LNP delivery technology provides strong support for the research and development of mRNA cancer vaccines.
Mechanism of Action of mRNA-LNP Cancer Vaccines
The administration of mRNA-LNP vaccines through intramuscular injection enables the body to produce target antigens which activate immune system responses. The vaccine that enters the body through intramuscular injection spreads throughout the lymph nodes. The mRNA-LNP complex uses cellular endocytic pathways including clathrin- and caveolin-mediated endocytosis and macropinocytosis to enter endosomes of dendritic cells (DCs). The protonated ionizable lipids enable mRNA to escape endosomal compartments and reach the cytoplasm where it gets translated into antigenic proteins inside DCs. The antigenic proteins become available to CD8+ T cells and CD8+ T cells through MHC I and MHC II pathways. The activation of CD8+ T cells results in CTLs which possess the ability to identify and destroy tumor cells. CD4+ T cells transform into Th cells which produce cytokines including interferon-γ (IFN-γ), tumor necrosis factor-α (TNF-α) and interleukin-2 (IL-2) to support and control immune system responses. B cells receive antigens from FDCs and Tfh cells which activate them for immune response. The germinal center process transforms these B cells into memory B cells and long-lived plasma cells. Plasma cells generate massive antibody production that helps defend the body against antigens through short-term immune responses.
Memory B cells persist for a long time, rapidly responding to re-exposure to the same antigen and providing long-term protection. The dual mechanism of mRNA-LNP vaccines enables them to generate strong cellular and humoral immune responses which effectively combat tumors. The antigen-induced immune response from mRNA also triggers innate immune system activation. When cells take up mRNA it becomes recognized as pathogen-associated molecular patterns (PAMPs) which bind to pattern recognition receptors (PRRs) on innate immune cells to activate their innate response. The immune system recognizes mRNA through activation of Toll-like receptors TLR3, TLR7 and TLR8 and other receptors. The innate immune response becomes activated through cytokine production of IFN which leads to better antigen-presenting cell activation and maturation and improved antigen presentation efficiency and adaptive immune response enhancement. The self-adjuvant property of mRNA vaccines makes them less dependent on adjuvants while improving their protective capabilities. The ionizable lipids in LNPs have been shown to activate innate immunity through IL-6 secretion which promotes germinal center B cell responses and Th cell and plasma cell and memory B cell activation. Research has shown that LNPs function as adjuvants to generate antigen-specific CD8+ T cells which results in tumor growth inhibition in mice and improved anti-tumor outcomes.
Key Technologies for mRNA Vaccines
The development of mRNA vaccine technology through key advancements during the last 30 years has improved their suitability for cancer immunotherapy applications. The combination of nucleoside modifications ψ and m1ψ with structural elements 5′ caps and polyadenylation tails and codon optimization has led to improved mRNA stability and translation efficiency and safety outcomes. Scientists have dedicated their research to developing antigen designs which utilize tumor-associated antigens (TAAs) and tumor-specific antigens (TSAs). The combination of different tumor-associated antigens and personalized tumor-specific antigens in vaccine formulations has proven successful for generating targeted T cell responses. Fusion antigen technology has further enhanced vaccine immunogenicity. In recent years, self-replicating mRNA (saRNA) and circular RNA (circRNA), as next-generation platforms, have become a new trend in mRNA vaccine development due to their enhanced expression capacity, longer expression duration, and reduced immunogenicity.
LNP Delivery System
LNPs (lipid nanoparticles) are currently the most widely used non-viral delivery system for mRNA vaccines. They have been successfully used in vaccines for various infectious diseases and also show promising prospects in cancer vaccine research. Traditional LNPs are composed of ionizable lipids, phospholipids, cholesterol, and PEGylated lipids. Microfluidic technology can be used to produce structurally stable and uniformly sized nanoparticles. Ionizable lipids are key components, possessing a positive charge in acidic environments to facilitate endosomal escape and remaining neutral in physiological conditions to reduce toxicity. Phospholipids contribute to particle stability and organ targeting, such as DOPE targeting the liver and DSPC targeting the spleen. The new iPhos series enables efficient organ-targeted delivery. Cholesterol enhances stability and delivery efficiency, and its derivatives, such as β-sitosterol and 7α-hydroxycholesterol, have shown excellent performance in enhancing transfection efficacy. PEGylated lipids prevent aggregation and clearance, but may trigger an immune response. Alternative materials, such as pSar, are currently being explored to reduce side effects. To solve the problem of mRNA-LNP accumulation in the liver in vivo, new technologies such as SORT can achieve precise delivery to tissues such as the lungs, spleen, and bone marrow by regulating the charge of LNPs; in addition, targeted LNPs based on lipid structure optimization and antibody modification are also contributing to breakthroughs in the delivery of mRNA vaccines to tumors and immune cells.
Clinical Progress of mRNA-LNP Vaccines
The first in vitro validation of mRNA cancer vaccines in dendritic cells occurred in 1996 but scientists have since made quick progress through mRNA technology and LNP delivery system advancements. Multiple mRNA-LNP cancer vaccines now participate in clinical trials at Phase I and II stages to assess their safety profile and initial treatment effectiveness and tolerance. Scientists now focus on developing personalized vaccines which use patient-specific genetic mutation information to create customized treatments. BioNTech launched BNT122 in 2023 for pancreatic cancer treatment because it sustained T cell activation and decreased relapse occurrences while starting Phase II clinical trials. The joint development of mRNA-4157 (V940) by Moderna and Merck achieved Phase IIb success in melanoma trials by lowering relapse and distant metastasis risks before entering Phase III trials. The development of mRNA-LNP cancer vaccines continues at a fast pace because they show promise to introduce individualized cancer treatment methods.
Related Services
The Development Process of mRNA-LNP Vaccine
mRNA-LNP Vaccine Laboratory Process Development Assay
Custom Innovative Ionizable Cationic Lipids for Safe and Efficient mRNA-LNP Formulation
